 New immune checkpoint inhibitors (ICIs) have been approved by the U.S. Food & Drug Administration and European Medicines Agency, and ICIs have demonstrated efficacy in the treatment of cancer, promoting overall survival and elongating the duration of remission. However, ICIs have also been associated with various autoimmune and inflammatory syndromes known as immune-related adverse events.
New immune checkpoint inhibitors (ICIs) have been approved by the U.S. Food & Drug Administration and European Medicines Agency, and ICIs have demonstrated efficacy in the treatment of cancer, promoting overall survival and elongating the duration of remission. However, ICIs have also been associated with various autoimmune and inflammatory syndromes known as immune-related adverse events.
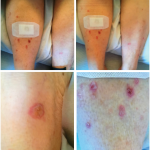
ICI-induced inflammatory arthritis, an immune-related adverse event, can occur during the beginning of ICI treatment and may persist for three to six months after ICI discontinuation. In some cases, inflammatory arthritis remains active for years after ICI cessation. Longer ICI exposure, combination ICI therapy and a history of other immune-related adverse events are all associated with an increase in the risk of persistent inflammatory arthritis.
The condition may require chronic treatment, which can present rheumatologists with the challenge of using immunosuppression to treat the arthritis while simultaneously being concerned the immunosuppression may reverse cancer remission. Unfortunately, lack clear guidance on the appropriate duration for treating this inflammatory arthritis is lacking, as is a thorough understanding of the effects of lengthy immunotherapy on specific immune-related adverse events. Rheumatologists recognize that most patients with ICI-induced inflammatory arthritis require systemic corticosteroids to treat the inflammatory arthritis and many require additional immunosuppression with conventional synthetic disease-modifying antirheumatic drugs (DMARDs) or biologic DMARDs, such as tumor necrosis factor (TNF) inhibitors and interleukin (IL) 6 inhibitors.
Tawnie J Braaten, MD, a rheumatologist at Johns Hopkins School of Medicine, Baltimore, and colleagues investigated the long-term outcomes of patients who develop ICI-induced inflammatory arthritis. They sought to define the factors associated with inflammatory arthritis persistence after ICI cessation, the need for immunosuppressants to treat inflammatory arthritis and the effect of these medications on underlying malignancies.
The investigators report that patients who develop ICI-induced inflammatory arthritis often experience long-term disease, requiring rheumatology care and immunomodulatory treatment. This treatment was not associated with cancer outcomes. The researchers published their findings in the March 2020 issue of the Annals of Rheumatic Disease.1
The Data
The study, one of the largest longitudinal reports of patients with ICI-induced inflammatory arthritis, included 60 patients (32 female and 28 male) referred to rheumatologists for joint complaints. It’s also the first study to evaluate the persistence of ICI-induced inflammatory arthritis and identify influencing factors.
The investigators monitored patients for a median follow-up of nine months after ICI cessation and an average follow-up of 12 months. The median baseline 28 swollen joint count was six, and the median baseline 28 tender joint count was two. The majority (75%) of patients required immunomodulatory treatment for their arthritis.