In a Phase 3 study, ixekizumab proved safe and effective to treat adults with ankylosing spondylitis (AS)…


In a Phase 3 study, ixekizumab proved safe and effective to treat adults with ankylosing spondylitis (AS)…

When Marc McClintock first began experiencing back pain, he chalked it up to the rigors of his racing career. For more than 36 years, Mr. McClintock has built and raced stock cars, high-powered race vehicles that compete on short oval or circular dirt or paved tracks. “I live in a commercial building, with my residence on…
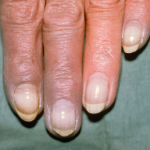
SAN DIEGO—Top researchers gathered for a review course at the start of the 2017 ACR/ARHP Annual Meeting in November to describe new research, their own treatment strategies and new ways of thinking about an array of rheumatic diseases. Here are the highlights: Raynaud’s & Other Digit Problems When a patient walks into your clinic with…

Innate lymphoid cells (ILCs) in the joints of patients may drive the pathology of spondyloarthritis through the production of granulocyte colony-stimulating factor (GM-CSF). A recent study found that both GM-CSF-producing CD4 T cells and the GM-CSF+ Th17 cells expressed high levels of GPR65—indicating that both GM-CSF and GPR65 may be therapeutic targets for spondyloarthritis…

Bisphosphonate Drug Holidays Drug holidays are common for patients on bisphosphonate therapy. Often, these breaks in treatment are related to known U.S. Food and Drug Administration (FDA) warnings and drug class adverse effects. Currently, data on fracture risk related to drug holidays are limited. In recent research highlighted at the 2017 ACR/ARHP Annual Meeting ,…
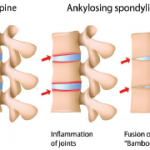
What factors drive inflammation and progressive disease in ankylosing spondylitis (AS)? The answers have long eluded rheumatologists. Although 90% of patients with AS test positive for the HLA-B27 gene, pieces remain missing in our understanding of this chronic, inflammatory disease, which often leads to pain, spinal fusion and, in about half of patients, gut involvement,…

In a Year in Review session at the 2017 ACR/ARHP Annual Meeting, Daniel Solomon, MD, MPH, highlighted the latest and most intriguing aspects of clinical research on rheumatic diseases from 2017. His discussion touched on medical therapy, genetics, the effects of bariatric surgery and diet, cancer risk and more…

Medications for serious or life-threatening conditions may receive accelerated approval from the U.S. Food and Drug Administration (FDA) by showing an effect on surrogate measures that are reasonably likely to predict a treatment’s clinical benefit. Post-approval confirmatory drug trials are then required to determine whether or not these effects translate into clinical improvements. In recent…

FDA Approves Adalimumab-adbm On Aug. 29, the FDA approved Cyltezo (adalimumab-adbm), a biosimilar to Humira (adalimumab).1 Cyltezo was approved as a prefilled syringe to treat multiple chronic inflammatory diseases, including moderate to severe active RA, active psoriatic arthritis and ankylosing spondylitis, and moderate to severe plaque psoriasis. The treatment has also been approved for moderate…

In an extension study, nearly half the patients with plaque psoriasis taking secukinumab maintained skin clearance for the five years of the study…