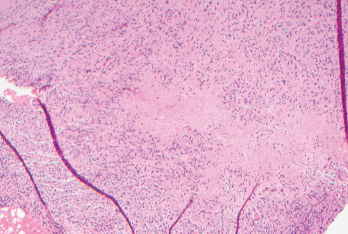
Figure 1. Biopsies of the small bowel mesenteric and serosal nodules demonstrated necrotizing granulomatous inflammation.
Common variable immunodeficiency (CVID) is a common primary immunodeficiency disease, with an estimated incidence of one per 25,000–50,000 individuals.1 The classic presentation includes frequent bacterial infections, secondary to dysfunctional B cell differentiation, impaired immunoglobulin production and diminished antibody response. The clinical presentation may be heterogenous and may include granulomatous disease as an uncommon manifestation. Granulomatous disease has an estimated incidence of 8–22% among CVID patients.2
Here, we describe a patient who presented with small bowel obstruction and was later discovered to have multiple mesenteric and small bowel serosal lesions on exploratory laparoscopy. Biopsies revealed necrotizing granulomas. The patient was eventually diagnosed with CVID following an extensive workup, and his symptoms resolved with intravenous immunoglobulin (IVIG) therapy.
Case Report
A 65-year-old man admitted for abdominal pain with hypotension was found to have small bowel obstruction. Imaging revealed soft tissue density mesenteric masses suspicious for carcinoid tumors. The patient was managed conservatively and was referred to surgical oncology for further evaluation.
The patient had a complex past medical history notable for atrial flutter, for which he underwent cardiac ablation; interstitial lung disease with associated pulmonary nodules, likely secondary to occupational exposure to coal dust and asbestos, as well as smoking; and a pancreatic head mass, which was incidentally discovered on surveillance imaging for his pulmonary nodules. Multiple fine needle aspirations of the pancreatic mass were nondiagnostic, and the mass eventually disappeared on imaging over the course of several months.
The patient underwent exploratory laparoscopy, which showed multiple mesenteric and small bowel serosal lesions. Biopsies of the lesions were negative for malignancy but showed necrotizing granulomatous inflammation (see Figure 1). The patient was subsequently referred to an infectious diseases specialist for further evaluation.
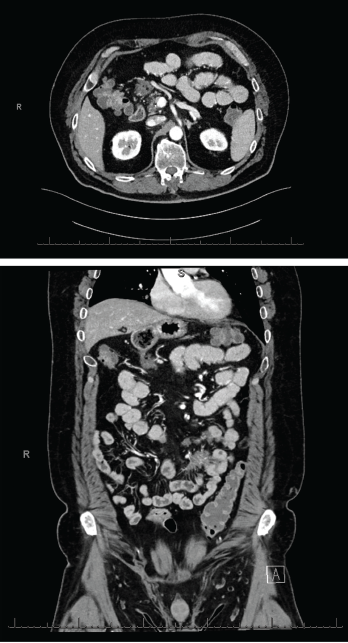
Figures 2 & 3 A CT enterography showed infiltrating soft tissue masses within the mesentery in a predominantly perienteric distribution.
An extensive infectious evaluation was unrevealing (see Table 1). Follow-up computerized tomography (CT) enterography showed similar soft tissue density infiltrating masses within the mesentery (see Figures 2 and 3). A CT scan of the chest showed multifocal, multilobar, subsegmental-sized opacities of unclear etiology (see Figure 4). These findings increased suspicion for an autoimmune etiology of his condition. The patient was referred to a rheumatologist to be evaluated for sarcoidosis, vasculitis and other rheumatic diseases.
At his rheumatology evaluation, the patient noted an unintentional 20 lb. weight loss over the previous year, which had since stabilized, and development of vitiligo. He also described nocturnal wheezing with exertional shortness of breath that had started more than 10 years before; those symptoms were treated with montelukast and a mometasone inhaler without clear benefit. The patient denied eye inflammation, chronic sinusitis, frequent infections, rash or joint pain. The physical exam was notable for vitiligo on his fingers, but was otherwise unremarkable.
The patient did not have anti-neutrophil cytoplasmic antibodies (ANCA). Inflammatory markers, angiotensin-converting enzyme and serum IgG4 were within normal limits. Immunoglobulin levels were decreased, with an IgG of 411 mg/dL, IgA of 49 mg/dL and IgM of 10 mg/dL. His CD19 B cell count was low, at 34 cells/μL, and his CD4+ T cell count was also low, at 350 cells/μL (see Table 1). These findings led to the diagnosis of CVID.
The patient was referred to an immunologist and initiated on 500 mg/kg IVIG every two weeks for two doses, followed by 500 mg/kg every four weeks. He was also treated with prophylactic sulfamethoxazole and trimethoprim (800 mg and 160 mg, respectively, daily), which was stopped several months into IVIG therapy.
A CT scan of the abdomen and pelvis, conducted three months after the patient was started on IVIG, showed resolution of the soft tissue density mesenteric masses. A CT scan of the chest, without contrast, showed a decrease in the multifocal, multilobar opacities, which were thought to represent granulomatous-lymphocytic interstitial lung disease (GLILD) in the setting of CVID.
Ten months following initiation of IVIG, the patient appeared clinically well. However, a repeat CT scan of the chest showed continued pulmonary disease, with interim resolution of several old pulmonary nodules, alongside the development of multiple new nodules. A pulmonary function test showed mild restrictive disease, with decreased diffusing capacity for carbon monoxide (DLCO) compared with 10 months before (forced vital capacity [FVC] 72%, compared with 78%; forced expiratory volume in one second [FEV1] 73%, compared with 79%; FEV1/FVC 75%, compared with 75%; and DLCO 91%, compared with 85%).
The patient underwent a bronchoscopy with endobronchial ultrasound and biopsy, as well as bronchoalveolar lavage. The biopsy showed reactive-appearing lymph node tissue and rare non-necrotizing granulomas. Acid-fast bacilli and Grocott methenamine silver stains were negative for mycobacterial or fungal infections.

