Working with more than 40 other medical associations, the ACR is supporting proposed federal legislation, the Local Coverage Determination Clarification Act of 2017 (S. 794). The bill is co-sponsored by Sens. Johnny Isakson (R-Ga.), Tom Carper (D-Del.), John Boozman (R-Ark.) and Debbie Stabenow (D-Mich.).
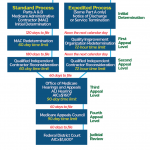
The bill’s provisions would add transparency to how Medicare Administrative Contractors (MACs) set or amend local coverage determinations (LCDs) for treatments or services reimbursed by Medicare. It would require MACs to hold open meetings, disclose evidence to back up their determinations, and provide ample time and opportunity for providers and suppliers to appeal decisions.
MACs & LCDs
MACs were created as a result of the 2003 Medicare Prescription Drug Improvement and Modernization Act (MMA), a law that reformed Medicare contracting. MACs are organizations that adjudicate Medicare Parts A and B claims and LCDs, which are decisions made by MACs on whether or not to cover medical items or services throughout their coverage area in accordance with Section 1862(a)(1)(A) of the Social Security Act.
“Medicare LCDs affect all patients and prescribers,” says Sean M. Fahey, MD, a rheumatologist in Mooresville, N.C., and the chair of the ACR’s Insurance Subcommittee. “The restructuring of the Medicare contractor responsibilities has given some MACs jurisdiction over large swaths of the country—some up to 10 states.” As a result, rheumatologists and other stakeholders have fewer opportunities to communicate with medical directors at MACs, which set local medical policies, or LCDs, he says. In addition, local coverage determinations often become national in scope because MACs can adopt each other’s draft LCDs and make them widespread policy.
“The LCD process has come into play for rheumatologists and our patients regarding coverage for injectable biologics and tests commonly ordered by rheumatologists, as well as delineation between medications that are considered complex and medications that are considered simple for infusion administrative purposes,” says Dr. Fahey.
Other Proposed Provisions: Transparency, Appeals
794’s proposed provisions would modify the process of creating or modifying LCDs to make it more transparent and inclusive of stakeholders’ input, says Dr. Fahey.
“It would create public meetings for the Contractor Advisory Committee and have minutes posted on the website for each MAC. It would require the MAC provide rational and preliminary evidence for why a new or revised LCD is being considered,” he says. This change would help prevent MACs collaborating on LCDs and making them “de facto national coverage determinations.”
The bill also proposes the creation of a process for providers and medical care suppliers to formally appeal the MAC’s decision to the Center for Medicare and Medicaid Services (CMS). Currently, CMS rules make MAC LCDs final, and do not allow review or appeal.