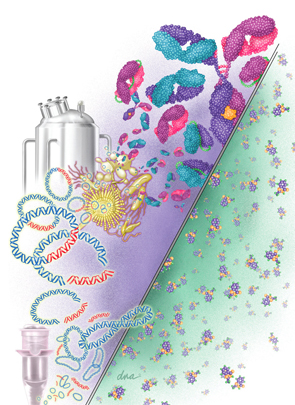
Biosimilars are much bigger and more complex on the molecular level than small-
molecule drugs. On the right is the small-molecule structure of aspirin (21 molecules). On the left is a monoclonal antibody made up of 10,000–20,000 molecules. The various biosimilar drugs depicted include monoclonal antibodies, scFv, Fab, and scFab, whereas the small-molecule drugs are identical. Also shown is a vial used for recombinant DNA procedures with plasmids as vectors and the desired fragments of DNA that are attached to the plasmids, forming recombinant plasmids. These are inserted into various living organisms, which produce the desired biosimilars in a fermentation tank.
DNA Illustrations/ScienceSource.com
On Feb. 9, the Arthritis Advisory Committee of the U.S. Food and Drug Administration (FDA) met in Washington, D.C., and recommended the approval of CT-P13, a proposed biosimilar to infliximab (Remicade), for indications including the treatment of rheumatoid arthritis, in combination with methotrexate; ankylosing spondylitis; and psoriatic arthritis. The human monoclonal antibody would be the first biosimilar available in the U.S. market to treat rheumatic diseases.
Biosimilars will offer welcome cost savings for both patients and payers. Still, some rheumatologists express concerns about the drugs, including how they will be named, fulfilled by the pharmacy, and tracked for safety and efficacy. The FDA has established two potential designations for these drugs—biosimilar and interchangeable—based on how their clinical data meet certain standards compared with the already-licensed “reference” biologic.
Sponsors must prove that the drugs are highly similar to the reference product through extensive, comparative analysis and a clinical study to assess meaningful differences between the two products. Biosimilars are biologics that are highly similar to the reference drug, but interchangeables must meet a higher standard and could be substituted for the reference drug by pharmacies without prescriber knowledge/intervention. An interchangeable must demonstrate that it produces the same clinical result as its reference product in any given patient and that the risk, in terms of safety, or diminished efficacy of alternating or switching between an interchangeable and the reference product is not greater than remaining on the reference drug. This means that the interchangeable drug may be substituted for the reference drug by a pharmacist without intervention by the prescriber. State laws govern pharmacy substitution, so it’s a complex, unfolding process.
The ACR Is Leading the Way
Rheumatologists have made biosimilars a high legislative priority to ensure that physicians’ voices are heard. Testifying on behalf of the American College of Rheumatology at the Feb. 9 meeting, Angus B. Worthing, MD, FACR, FACP, a rheumatologist practicing in Chevy Chase, Md., expressed the need for these lower-cost treatments.
“As more biologic drugs are used to treat rheumatic diseases, rheumatologists are increasingly concerned about their high costs and patients being able to afford them,” said Dr. Worthing, a member of the ACR Government Affairs Committee. “The ACR strongly believes that safe and effective treatment alternatives should be made available to patients at the lowest possible cost.”
CT-P13 is a biosimilar monoclonal antibody manufactured by Korea-based Celltrion. Called Remsima outside the U.S., it was approved by the European Medicines Agency (EMA) in 2013 and is also approved in Canada and Japan. Remicade, which was first approved by the FDA in 1998, is owned by Janssen Biotech Inc. and is used to treat rheumatoid arthritis, ankylosing spondylitis and psoriatic arthritis.
It won’t be the first biosimilar on the U.S. market—filgrastim-sndz (Zarxio), a cancer drug approved by the FDA in March 2015, has that distinction—but it’s an important milestone on the road to more affordable rheumatologic treatments, said David Daikh, MD, PhD, Kenneth H. Fye Chair in Rheumatology at the University of California, San Francisco.
“The potential for cost savings for patients and the availability of more cost-effective, effective treatment options are the obvious, hoped-for benefit of these drugs,” said Dr. Daikh. This year, companies that make biosimilars to etanercept (Enbrel) and adalimumab (Humira) are expected to seek FDA approval.
‘As more biologic drugs are used to treat rheumatic diseases, rheumatologists are increasingly concerned about their high costs & patients being able to afford them. The ACR strongly believes that safe & effective treatment alternatives should be made available to patients at the lowest possible cost.’ —Dr. Worthing, member of the ACR Government Affairs Committee
Because they are complex molecules that depend on living cells, biosimilars aren’t as simple to manufacture as small-molecule generic drugs. They require comprehensive testing of their immunogenicity to ensure safety. Even small changes can have major biologic effects that can potentially impact efficacy or put patients at serious risk, said Dr. Daikh.
“My major concerns are that as biosimilars are developed, there is adequate study to determine how closely they approximate their index agent, and that they are not automatically substituted as ‘generics’ because of cost or formulary preference,” he said. Rheumatologists need assurance that biosimilar use will be specifically tracked so that problems that might only turn up late and after more extensive experience can be identified, just as is done for conventionally approved unique drugs.
Seemingly minor changes in formulation can cause major adverse effects. When a stabilizing agent in Eprex, a biosimilar of epoetin alfa used for treating anemia that’s approved outside the U.S., was changed, it led to pure red cell aplasia (PRCA) in some patients.1
“It’s very important to understand that a biosimilar agent will never be identical, at a molecular level, to its reference drug,” said Douglas W. White, MD, a rheumatologist at Gundersen Health System in Onalaska, Wis. “Variations inherent in the manufacture of biosimilars can result in highly divergent responses in humans, especially with respect to immunogenicity, as we saw with Eprex. So we want safeguards in place to minimize such a possibility and to maximize our chances of catching it early should something go wrong.”
Although Dr. Daikh has confidence that pharmaceutical companies with experience in developing biologics will maintain high-quality control, “it will be incumbent upon the FDA to ensure that all biosimilars are manufactured to the same quality and purity standards applied to the parent drugs, regardless of where and by whom the biosimilars are produced.”
The ACR strongly suggests the following standards for biosimilars, Dr. Worthing testified:
- Long-term post-marketing studies of approved biosimilars to monitor for adverse events;
- Distinct names for each biosimilar to ensure correct prescribing and dispensing, as well as monitoring and tracking; and
- Clear FDA labeling that indicates whether a drug is: a) a biosimilar or interchangeable product; b) lists all approved indications for the biosimilar; and c) specifies whether clinical data come from studies of the biosimilar or the reference biologic.
Currently, U.S. biosimilars will each have a four-letter suffix to identify their reference biologic, instead of a distinct brand name. Some rheumatologists fear this naming style will cause confusion.
“We need to be able to confidently prescribe these therapies to our patients and feel comfortable that proper mechanisms are in place for rheumatologists to monitor patient reactions to prescribed biosimilars,” said Dr. Worthing. “Rheumatologists have been working with biologics for quite some time, and our patients take them for lengthy periods of time to manage their conditions, so we have a unique understanding of the extreme complexities of these medications.”
The pharmaceutical industry also wants the new products to have memorable, meaningful names, said Geoff Eich, executive director, Amgen Biosimilars.
“It will be more accurate in the database if you can actually remember it and track any adverse events to the appropriate product,” said Mr. Eich. He’s optimistic that biosimilars will encourage robust competition that will fuel innovation. “There’s opportunity for biosimilars around the world to be very successful.”
Pathway to Approval
Biologics are expensive to produce and costly for payers and many patients, depending on their insurance coverage. Adalimumab and etanercept have retail costs of $3,000 and higher per monthly dose, although coupon programs can reduce out-of-pocket costs for some patients.
According to a report by the Centers for Medicare & Medicaid Services (CMS), 22 of the top 48 brand-only drugs covered by Medicare Part B coverage are biologics.
The costs of biologics have been rising steadily, year over year. For example, wholesale acquisition prices for two top biologics used to treat RA, adalimumab and etanercept, increased 9% annually from 2007 to 2012, according to a study by Prime Therapeutics, a pharmacy benefit manager, presented at the Academy of Managed Care Pharmacy Annual Meeting in April 2013. The study projected that if cost increases continued at the same rate each year, RA specialty drug costs would likely top $50,000 per person by 2022.2
According to CMS’ Medicare Drug Spending Dashboard report for 2014, the annual change in average cost per unit from the prior year for adalimumab was 15%, etanercept 16% and infliximab 5%.
To slow these upward trends and encourage the availability of more cost-effective treatments, Congress passed the Biologics Price Competition and Innovation Act, part of the Affordable Care Act (ACA), in 2009. This created a new, possibly faster approval pathway for products that are biosimilar to or interchangeable with an FDA-licensed reference product.
“The FDA is committed to encouraging the development of these biosimilar and interchangeable products,” said Leah Christi, PhD, the FDA’s associate director for therapeutic biologics.
Biosimilars may have a shorter, less costly path to approval because their sponsors can rely on existing scientific knowledge of the reference biologic to support safety and efficacy, she said. This quicker route to market “is one way to improve access and increase treatment options for the public and potentially lower cost.”
Despite being able to rely on existing data, the FDA will apply the same high standards for approving biosimilars, said Dr. Christi. This includes a totality-of-the-evidence approach, considering all available data, including analytical, animal, human pharmacokinetics, human pharmacodynamics when applicable, clinical immunogenicity, and clinical safety and effectiveness.
What the 2009 law requires is that sponsors demonstrate that these products are “highly similar to the reference product, notwithstanding minor differences in clinically inactive components,” and that “there are no clinically meaningful differences between the biological product and the reference product in terms of safety, purity and potency of the product,” said Dr. Christi.
What about extrapolation of data so a biosimilar can be approved for the same indications as its reference product? That’s one unknown that concerns some rheumatologists, said Dr. White. The ACR is closely engaged with the FDA to ensure that “clinical data from one population of patients are not extrapolated to another patient population, thus allowing FDA approval in an untested population without great caution,” he said.
Although the FDA’s biosimilar pathway allows for clinical data to be extrapolated to additional indications, applicants will still need to provide adequate scientific justification for doing so, said Dr. Christi.
To give prescribers accessible information about these new drugs’ designations and indications, the FDA has published an online reference, The Purple Book, which lists licensed biologics with reference product exclusivity, and under these, the biosimilars and interchangeables for them.
Control & Tracking
Clear communication and naming of these drugs will help rheumatologists and others track efficacy, said Dr. Daikh.
Without distinct naming, “it will be difficult to tell if a patient has been switched, and to sort out whether a clinical change resulted from such a switch. It will also be impossible to track aggregate experience with the drugs, such as through adverse drug event reporting, to learn about potential safety signals with these agents,” he said.
No applications have been made yet for interchangeables, but pharmacists would be able to substitute these drugs for the reference biologic without notifying the prescriber. Seventeen states passed laws to require notification anyway, and more are considering legislation.
Pharmacists want to lower costs, streamline fulfillment and work with prescribers to ensure safety, said Bernadette Eichelberger, PharmD, program director of the Academy of Managed Care Pharmacy’s Biologics and Biosimilars Collective Intelligence Consortium. This research network was founded in 2015 to conduct analyses on biosimilars and their reference biologics, using data collected from managed care organizations, pharmacy benefit managers and physicians.
“Our job is to ensure that when these drugs come to market, they are being monitored,” said Dr. Eichelberger. “If we don’t have active surveillance of these products, it puts patients at risk.”
Some patients may be required to take a biosimilar by their insurer if they’re prescribed a biologic that has one, said Mr. Eich. Rheumatologists will still have treatment options.
“Even in that instance, you will ideally have a significant clinical choice as to which biologic to use,” said Mr. Eich. “You may still maintain the decision rights around which product you prescribe.” However, a right to prescribe a product, does not mean that the insurer can’t insist on switching to a formulary biosimilar.
How Much Savings?
It’s hard to predict the financial savings of biosimilars until they come to market. By looking at past biologic sales, potential discounting due to increased competition, European biosimilar prices and increasing penetration of the drug market over the next decade, cost savings may top $44 billion over the next decade, and biosimilar price discounts may be as much as 35% lower, according to a 2014 study released by the RAND Corp.
Patients like Carolyn Amisano, who’s had RA for 42 years, say lower prices will make drugs accessible to more people who need them. “The price will make a real difference in whether you’d be able to take that medicine. If they can come out with something more affordable, it would be great for many people, and saving a lot of families,” said Ms. Amisano, who has used two biologics in the past. “It’s a shame if you have to let drug costs influence your health. As long as I know it will work, and if it’s going to do the same thing, then it would be tremendous.”
Although patients like Ms. Amisano are used to generic drugs, biosimilars won’t be as simple a switch, said Martin Zagari, MD, vice president of global health economics at Amgen, the biopharmaceutical company based in Thousand Oaks, Calif. Communication about biosimilars will help allay fears and confusion.
“We believe even more expertise will be required from such groups as the ACR. We should expect that most people are predicting that this notion of precision medicine and personalized medicine actually increases the role of deep expertise and patient-centeredness,” he said.
Room for Innovation
Savings generated by biosimilars will also open space in research and development budgets for future breakthrough products, said Dr. Zagari. “The medicines that are now seen as the most expensive are also going to be the ones that are creating new headroom for treatment. When these molecules are able to be provided in biosimilar form, they will have an effect on the system as profound as generic drugs have had to date.”
Focus on the improved, long-term health outcomes that more affordable biologic drugs can provide and the savings they will offer, he said.
“Predictability of innovation is actually becoming more and more something that we can count on as a society. How can we afford new medicines? In the 1980s and 1990s, only about one-third of medicines were generic. Now, it’s almost 90% of prescriptions. This is how we get [healthcare costs] under control,” he said.
The ACR leadership has done a tremendous job ensuring that biosimilars will work for clinicians and their patients, said Mr. Eich.
“The interest of society is having patients and physicians tell the industry, regulators, others: Here is how biosimilars can actually improve patient care, so patients can benefit and the practice of healthcare is improved,” he said.
Rheumatologists have been at the forefront in the use of targeted biologics and have deep experience to apply to ensure the safety and efficacy of biosimilars for their patients, said Dr. Daikh.
“It’s important that rheumatologists engage in this issue as biosimilars are being developed and as fundamental regulations regarding their production, testing and labeling are being promulgated,” he said.
Susan Bernstein is a freelance medical journalist based in Atlanta.
Terms to Know
Biosimilar: Biological therapy proved to be highly similar to an FDA-approved biological product, known as a reference product. Has no clinical, meaningful differences from the original drug in safety and effectiveness. Minor differences in clinically inactive components are acceptable.
BPCI: Biologics Price Competition and Innovation Act. Passed in 2009 as part of the Patient Protection and Affordable Care Act, created an abbreviated approval pathway for biologic drugs proved to be either highly similar to or interchangeable with an approved biologic product.
Core name: Non-branded name of originator, reference biologic. All products related to this originator will have the same core name, but may have a different, identifying suffix.
Interchangeable: Biological therapy that’s biosimilar to an FDA-approved reference product, but also meets additional standards and may be substituted for the reference product by a pharmacist without the prescriber’s intervention.
Proper name: Non-branded name of biologic drug that reflects its chemical structure or pharmacologic properties.
Proprietary name: Trademarked, brand name of a drug registered for private use by its manufacturer.
Purple Book: Online reference created by the FDA to list biosimilars and interchangeables under their reference products. Similar to existing Orange Book.
Reference product: Original, approved biologic therapy with which a biosimilar or interchangeable product may be substituted. No previously licensed biological product is related to this originator.
Suffix: Four-letter addition to the end of the core name of the biosimilar or interchangeable. The FDA proposes that suffixes be unique, non-promotional and not too similar to those of other products. These suffixes are designed to reduce inadvertent substitution, but it’s not yet confirmed whether an interchangeable will or will not have a unique suffix or will have the same proper name as the reference product.
References
- Schellekens H. Biosimilar therapeutics: What do we need to consider? NDT Plus. 2009;2(Suppl 1):i27–i36.
- Gleason P. RA drug therapy projected to exceed $50k per person in 2022. Prime Therapeutics. 2013 May 23.



