MOLEKUUL / Science Source
Drug-induced lupus erythematosus and ANCA-associated vasculitis (AAV) are both autoimmune conditions associated with the use of hydralazine, a commonly prescribed drug for hypertension and congestive heart failure. Although the pathogenesis is unknown, it is believed that hydralazine alters neutrophil and lymphocyte function and promotes exposure of antigens, leading to the development of anti-neutrophil antibodies (ANCA) and/or anti-nuclear antibodies (ANA) and ultimately triggering a systemic response.1
These conditions are rare and typically considered to be mutually exclusive. Here, we describe a rare presentation in which hydralazine appears to be the inciting factor of a patient developing an overlap of drug-induced lupus erythematosus and AAV.
Case Report
A 75-year-old Caucasian woman with a history of hypertension was admitted for fever, periorbital edema, erythematous conjunctiva, papular lesions all over her body and a mucosal ulceration on the hard palate of her oral cavity.
She had no reported history of Raynaud’s phenomenon, alopecia, photosensitivity, joint pain or synovitis. She denied a history of venous thromboembolism or recurrent pregnancy loss. She denied epistaxis and new-onset neuropathy. She had no recent changes to her medication, but had been using hydralazine 25 mg twice daily as the drug of choice for hypertension for more than a year. Her personal surgical and family history were otherwise unremarkable.
During her hospitalization, infection was ruled out and ocular findings were determined to be consistent with episcleritis.
Laboratory data revealed proteinuria and hematuria on initial urinalysis. Serum creatinine was elevated from her normal baseline at 1.44 mg/dL (normal: 0.50–1.40 mg/dL) with a BUN (blood urea nitrogen) level of 39 mg/dL (normal: 7–22 mg/dL). Her urine creatinine was 86.8 mg/dL and urine protein was 335 mg/dL, with a ratio of 3.86.
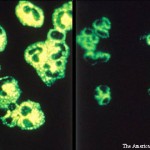
The complete blood count revealed leukopenia 3.6 cells/µl (normal: 4.5–10 cells/µl), and lymphopenia 10.1% (normal: 20–40%). An autoimmune evaluation was remarkable for ANA (1:640 speckled), anti-double-stranded DNA antibodies (1:10), myeloperoxidase (MPO) ANCA, and P-ANCA 1:320. Her C3 level was 69 mg/dL (normal: 88–201 mg/dL) and C4 was 10 mg/dL (normal: 16–44 mg/dL). Her C-reactive protein was elevated at 214 mg/L (<2.9 mg/L), as was the sedimentation rate at >100 mm/Hr (normal: 0–20 mm/hr). Her anti-histone immunoglobulin (Ig) G was 5.0 (normal: 0.0–0.9 units). Anti-Smith, anti-RNP and anti-SSA/SSB antibodies were negative. Antiphospholipid antibodies were negative. Cryoglobulin, anti-glomerular basement membrane antibodies and proteinase-3 (PR3) ANCA were undetectable.
CT imaging revealed stenosis at the left subclavian artery, multiple sub-centimeter pulmonary nodules, cardiomegaly and trace pericardial and pleural effusions. Additionally, incidental findings of hepatic and renal cysts were noted.