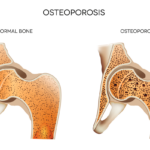
NEW YORK (Reuters Health)—Gastrointestinal side effects are the most common reason osteoporotic women cite for nonadherence to oral bisphosphonate therapy, according to a new survey.
“Our findings highlight the importance of low tolerability to nonadherence with osteoporosis therapy and underlines patients’ poor awareness and suboptimal physicians’ involvement in conveying the importance of this therapy,” Dr. Inbal Goldshtein of Maccabi Healthcare Services in Tel Aviv and colleagues state in their report, published on online June 21 in Advances in Therapy.
Although bisphosphonates reduce fracture risk in women with osteoporosis, adherence to and persistence with the medications is poor, the researchers write. Several new treatment options, as well as “rare, but alarming” side effects, such as osteonecrosis of the jaw and atypical femur fractures, “have influenced both physicians and patients, and may cause intentional non-adherence with the initially prescribed medication,” they add.
To better understand factors involved in nonadherence or choices to switch treatment, the researchers conducted a survey of 493 women (mean age 66) newly prescribed risedronate or alendronate in 2010–2012 who were either nonadherent (medication possession ratio less than 70%) or changed to a different therapy within a year.
Forty percent stopped taking any anti-osteoporotic treatment, 9% continued on the initial therapy and 51% switched to a different therapy. Among those who discontinued or switched therapy, 40% cited heartburn, acid reflux or other gastrointestinal side effects and 26.7% cited physician recommendation. Patients who discontinued therapy completely most commonly cited side effects (26.9%) and physician recommendation (20%), while 14% cited perceived low importance and 3% cited medication cost.
“Our findings mainly emphasize the need for new medications with a better tolerability profile, and the physician’s unfulfilled role in conveying the benefits of therapy,” Dr. Goldshtein and colleagues write.
“Physicians should further experiment with the various potential strategies to promote adherence, including patients’ education regarding the necessity, expected effect, and duration of treatment, as well as patients’ involvement in choosing the preferred medication regimen,” they suggest.
The authors were unavailable for comment. Merck & Co., which makes alendronate, funded the study and employed one of the coauthors.