 ACR CONVERGENCE 2021—Hypereosinophilic syndromes (HES) include a wide array of rare diseases characterized by peripheral eosinophilia and end-organ involvement. Because eosinophilic granulomatosis with polyangiitis (EGPA) occupies the space between HES and vasculitis, rheumatologists are often tasked with diagnosing and managing other HES, as well. Amy D. Klion, MD, chief of the Human Eosinophil Section, U.S. National Institutes of Health and National Institutes for Allergy and Infectious Diseases (NIH/NIAID), Bethesda, Md., provided a detailed overview of the EGPA and HES spectrum at ACR Convergence 2021, Nov. 3–9, 2021.
ACR CONVERGENCE 2021—Hypereosinophilic syndromes (HES) include a wide array of rare diseases characterized by peripheral eosinophilia and end-organ involvement. Because eosinophilic granulomatosis with polyangiitis (EGPA) occupies the space between HES and vasculitis, rheumatologists are often tasked with diagnosing and managing other HES, as well. Amy D. Klion, MD, chief of the Human Eosinophil Section, U.S. National Institutes of Health and National Institutes for Allergy and Infectious Diseases (NIH/NIAID), Bethesda, Md., provided a detailed overview of the EGPA and HES spectrum at ACR Convergence 2021, Nov. 3–9, 2021.
Hypereosinophilic Syndromes
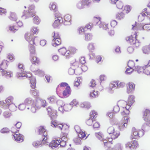
HES is an umbrella category for several clinical subtypes. In the NIH cohort, greater than 50% fall into the idiopathic HES (iHES) category.
“Despite extensive evaluation, no apparent etiology is identified, and the clinical picture does not fit one of the defined subtypes,” Dr. Klion said. “Further, a subset of patients [with an idiopathic hypereosinophilic syndrome] is completely asymptomatic and have no evidence of end-organ involvement.”1
Overlap HES is also common. Example: A single-organ eosinophilic disorder, such as chronic eosinophilic pneumonia, may overlap with iHES, or EGPA may overlap with HES. “This is important to recognize [because] the therapeutic approach may be different,” she said.
Marked eosinophilia may also occur in a variety of myeloid neoplasms and myeloproliferative disorders. Platelet-derived growth factor receptor-α (PDGFRα) associated myeloid neoplasm is the most common in this category. It’s caused by an interstitial deletion in chromosome 4 that leads to a constitutively activated fusion tyrosine kinase. Fortunately, this variant is sensitive to imatinib. Before imatinib, three-year mortality was 30%.2
EGPA
Although most rheumatologists think of EGPA as separate from HES, the signs and symptoms are similar. Anti-neutrophil cytoplasmic antibodies (ANCA) are positive in less than 50% of patients with biopsy-proven EGPA, so we cannot rely on ANCA testing alone to differentiate the two. EGPA and HES with asthma or sinusitis show overlapping serum biomarker profiles, so we cannot rely on this profile to separate them either.3
“So are EGPA and HES a spectrum or an overlap?” Dr. Klion asked.
Recent studies have shown that ANCA-positive EGPA patients are more likely to have neurologic involvement, renal involvement and purpura, but less likely to have cardiac involvement. Relapses are more common in the ANCA-positive group, but mortality is increased in the ANCA-negative group.4,5
How should we approach ANCA-negative EGPA patients who don’t fall into a neat little box? Dr. Klion said, “Most of the patients with EGPA/HES overlap are ANCA negative. So when we have an HES patient who has features of EGPA without biopsy-proven vasculitis, we treat them the way the 2021 ACR/Vasculitis Foundation Guideline for the Treatment and Management of ANCA-Associated Vasculitis would recommend you treat EGPA. But if they don’t respond to this approach, we try another agent that works for both, [such as] mepolizumab. Fortunately, mepolizumab is now FDA approved for the treatment of both HES and EGPA, so insurance approval has gotten easier.”6
Next Steps
Dr. Klion closed with thoughts on next steps in the treatment of these conditions. “Both represent part of the larger umbrella of eosinophilic syndromes. Understanding better the pathogenesis of each—particularly the way in which ANCA may be involved—will be imperative for deciding which targeted therapy may be best. Other targeted therapies are under investigation and hopefully will be options in our armamentarium the near future,” she said.
Ask the Eosinophil Expert
The audience had many questions for Dr. Klion at the end of her talk. She graciously shared her expert knowledge.
Regarding bone marrow biopsies, she said, “We typically do a biopsy in any patient who has an absolute eosinophil count [AEC] of greater than 5,000, or any other markers that could suggest a neoplastic variant. Abnormal peripheral flow cytometry, elevated serum vitamin B12 or serum tryptase levels, or splenomegaly would warrant a biopsy, for example. In many ways, it’s really a very safe procedure, and it makes the patient feel more comfortable that there is nothing going on underneath.”
Regarding infectious evaluations, she said, “There is no specific infectious panel I send for work-up, … and I’m an infectious disease parasitologist. The one caveat I will note is that Strongyloides is worldwide, so I do think about it in every patient. If there is any risk they may be infected, U.S. Centers for Disease Control and Prevention [CDC] serologies are what we use. The commercial tests are not as good. If there is any suspicion, I would treat with ivermectin. Any other parasite testing would really require the right clinical history.”
Regarding other specialists and treatment, Dr. Klion said, “It’s important to remember eosinophils can cause the same clinical presentation no matter what the underlying cause is, and you should involve other specialists if the picture doesn’t fit into yours. Treatment should depend on the presence of end-organ involvement, and not the AEC alone.”
Summary
HES and EGPA represent a spectrum of eosinophilic disorders. Further studies are necessary to differentiate best treatment options for each, but our armamentarium of available options is growing.
Samantha C. Shapiro, MD, is an academic rheumatologist and an affiliate faculty member of the Dell Medical School at the University of Texas at Austin. She received her training in internal medicine and rheumatology at Johns Hopkins University, Baltimore. She is also a member of the ACR Insurance Subcommittee.
References
- Chen YYK, Khoury P, Ware JM, et al. Marked and persistent eosinophilia in the absence of clinical manifestations. J Allergy Clin Immunol. 2014 Apr;133(4):1195–1202.
- Cools J, DeAngelo DJ, Gotlib J, et al. A tyrosine kinase created by fusion of the PDGFRA and FIP1L1 genes as a therapeutic target of imatinib in idiopathic hypereosinophilic syndrome. N Engl J Med. 2003 Mar 27;348(13):1201–1214.
- Khoury P, Zagallo P, Talar-Williams C, et al. Serum biomarkers are similar in Churg-Strauss syndrome and hypereosinophilic syndrome. Allergy. 2012 Sep;67(9):1149–1156.
- Chang HC, Chou PC, Lai CY, et al. Antineutrophil cytoplasmic antibodies and organ-specific manifestations in eosinophilic granulomatosis with polyangiitis: A systematic review and meta-analysis. J Allergy Clin Immunol Pract. 2021 Jan;9(1):445–452.e6.
- Comarmond C, Pagnoux C, Khellaf M, et al. Eosinophilic granulomatosis with polyangiitis (Churg-Strauss): Clinical characteristics and long-term follow-up of the 383 patients enrolled in the French Vasculitis Study Group cohort. Arthritis Rheum. 2013 Jan;65(1):270–281.
- Chung SA, Langford CA, Maz M, et al. 2021 American College of Rheumatology/Vasculitis Foundation guideline for the management of antineutrophil cytoplasmic antibody–associated vasculitis. Arthritis Rheumatol. 2021 Aug;73(8):1366–1383.