Rawpixel.com / shutterstock.com
The past five years have been busier than usual for the Churg-Strauss syndrome. It was renamed eosinophilic granulomatosis with polyangiitis (EGPA).1 Longitudinal cohorts totaling 484 patients—approximately as many as all previous series combined—were described.2,3 A proposal was advanced to remove and rename a subset in which vasculitis may not be present.4 And shortly after the production of consensus guidelines for management, the portion of the evidence base comprising relatively large, randomized trials more than doubled.5
The Long & the Short of It
EGPA is a good name—and manageable once shortened to four letters. It describes the definitive pathology well. Just as with granulomatosis with polyangiitis (GPA, Wegener’s), EGPA does not feature granulomatous vasculitis, nor are eosinophils essential to the process of vasculitis. Rather, the eosinophilic granulomas are nearby, hence “with.” GPA is no longer the only rheumatic disease with a “with” in the name.
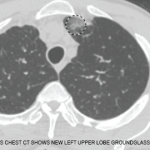
Churg-Strauss is an easier name to remember, but it doesn’t help physicians remember the clinical features, nor does it help patients educate their family and friends. What isn’t captured in the new name is that many features of the disease are characterized by eosinophilic inflammation into tissues, without necessarily being accompanied by vasculitis or necrotizing granulomatous inflammation. As far as can be determined, the pathology of nasal inflammation is indistinguishable from that of isolated nasal polyps; the parenchymal lung pathology is often the same as isolated eosinophilic pneumonia; and the eosinophilic myocarditis is indistinguishable from that seen in hypereosinophilic syndromes.
Sometimes 100 Is a Lot
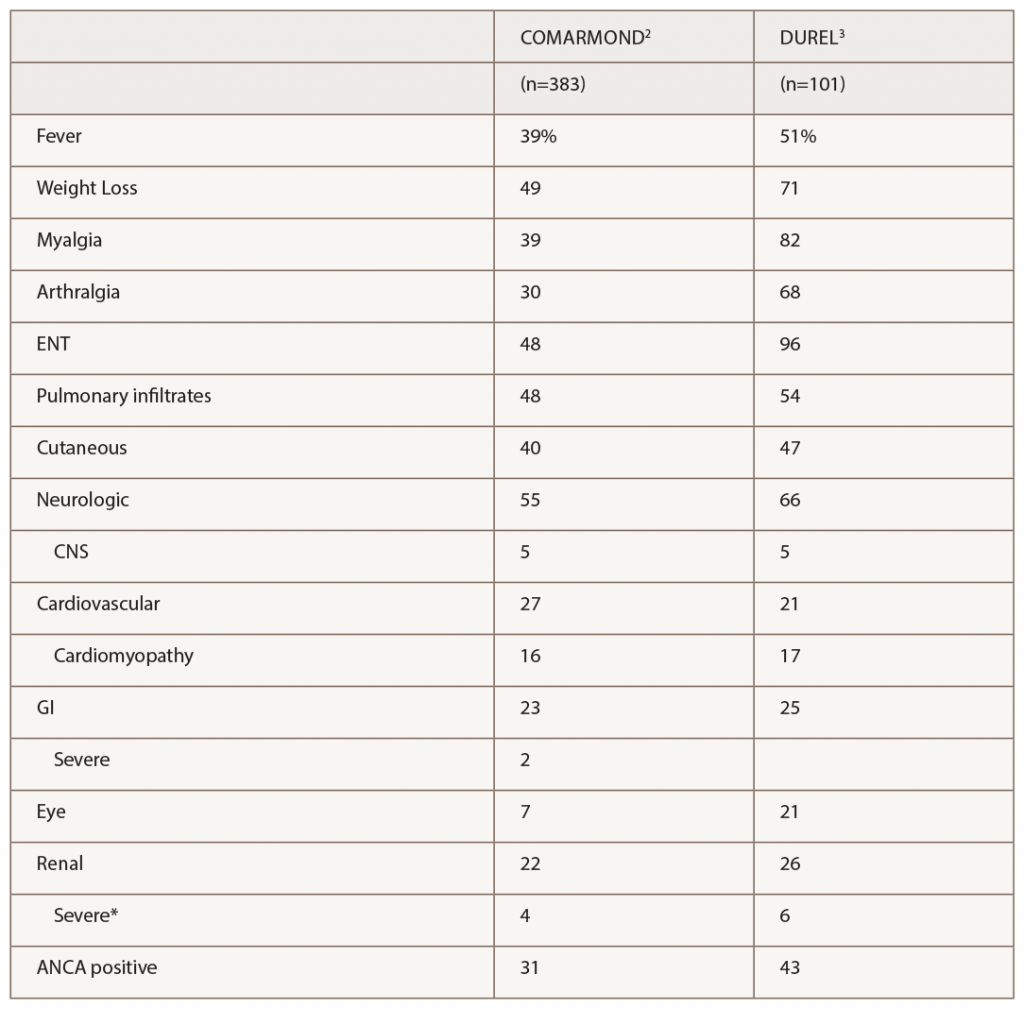
(click for larger image) Table 1: Manifestations of EGPA in 2 Recent Observational Cohort Studies
* Severe renal disease was defined by Comarmond as meeting Five Factor Score criteria for reduced renal function or proteinuria, and by Durel as having evidence for glomerulonephritis.
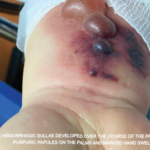
The series by Comarmond and the French Vasculitis Study Group (FVSG), and by Durel, provide useful details on clinical manifestations. A summary of organ system involvement is shown in Table 1, but readers are advised to look in the papers at the diversity of manifestations within a given organ system. For example, in the FVSG study, cutaneous manifestations (present in 40% of patients at diagnosis) are subdivided into purpura (23%), urticarial lesions (10%), subcutaneous nodules (10%), livedo reticularis (4%) and necrotic/gangrenous lesions (4%). Kidney involvement was surprisingly high (22–26%), but severe involvement was much less common (4–6%).2,3 Consistent with previous studies, 31–43% of patients were ANCA positive, and this was the group at risk for GN.3 Also consistent with previous studies, ANCA-positive patients were more likely to have peripheral neuropathy and ANCA-negative patients were more likely to have cardiomyopathy, although differences were not nearly as large as with GN.