Vladimka production / shutterstock.com
More than 7 million doses of the Johnson & Johnson single-dose vaccine have been administered in the U.S.1 On Apr. 23, the Centers for Disease Control & Prevention (CDC) and the U.S. Food & Drug Administration (FDA) recommended lifting the recommended pause on Johnson & Johnson COVID-19 vaccine use following a thorough safety review. 2 But healthcare providers should warn patients of the risk of developing the rare and serious blood clots that led to a pause in the vaccine distribution.
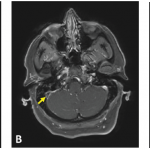
On April 13, the CDC and the FDA had recommended pausing the distribution of the Johnson & Johnson vaccine in the U.S. due to six reported cases of a rare and severe type of blood clot in the brain.3,4
All six cases of cerebral venous sinus thrombosis (CVST) and thrombocytopenia (i.e., platelet counts <150k/mm3) occurred in women between the ages of 18 and 48, with symptoms occurring 6 to 13 days after vaccination. All were hospitalized and admitted to the intensive care unit. One woman died.
The key reason for the pause was to alert the healthcare provider community of the potential for these adverse events, so they can recognize and appropriately manage this specific clotting issue. The treatment for CVST differs from the treatment that might typically be administered for blood clots: Heparin is often used to treat blood clots, but the administration of heparin in this setting may be dangerous and alternative treatments need to be given. A possible explanation could be the vaccine triggering an immune response similar to heparin-induced thrombocytopenia.4
The Advisory Committee on Immunization Practices (ACIP), an independent panel that advises the CDC on vaccine safety and administration issues, convened an emergency meeting on April 14 to discuss the pause recommendation.6 ACIP did not make a recommendation, which meant the temporary pause would stay in place and allow for a complete risk assessment and additional time to review additional cases. The rare events are only showing up now—with the reporting rate of 0.87 cases per million doses administered. The incidence of CVST associated with thrombocytopenia is unknown and is considered by the FDA and the CDC to be extremely low.5
The Johnson & Johnson vaccine is notably different from the Moderna and Pfizer COVID-19 vaccines in three ways: It is adenovirus-based, given as a single dose and can be stored for long periods of time in a refrigerator. The Moderna and Pfizer vaccines are made using messenger RNA (mRNA), and require two doses and an elaborate cold chain (i.e., an uninterrupted series of refrigerated production, storage and distribution activities, along with associated equipment and logistics, which maintain quality via a desired low-temperature range). They can be stored for just two weeks at temperatures of normal pharmacy freezers, between –13º and 5ºF.
With 182 million mRNA COVID-19 doses administered, no instances of CVST with thrombocytopenia have been observed with the two authorized mRNA vaccines. However, three cases of CVST have been reported to the Vaccine Adverse Event Reporting System (VAERS) with the Moderna COVID-19 vaccine.
The clinical features of the Johnson & Johnson cases are similar to those observed following administration of the AstraZeneca vaccine (Vaxzeria) in Europe. Both Johnson & Johnson and AstraZeneca vaccines contain replication-incompetent adenoviral vectors (human [Ad26.COV2.S] for Johnson & Johnson and chimpanzee [ChAdOx1] for AstraZeneca. The European Medicines Agency (EMA) never recommended a pause for Vaxzevria despite many more cases of the serious side effect, saying benefits outweigh risk.4
In a report published in The New England Journal of Medicine (NEJM), researchers from Johnson & Johnson disputed claims of classwide safety issues with adenoviral vector COVID-19 vaccines—that a class effect may be to blame for rare blood clots seen in some people who received Ad26.COV2.S.7 Although both Ad26.COV2.S and Vaxzeria use adenoviral vector platforms, the researchers point to differences, such as different vectors and spike (S) protein inserts, along with different host cell receptors that make it possible for the vaccines to have different biologic characteristics.
As concerns increased over rare cases of CVST with thrombocytopenia, Johnson & Johnson reached out to AstraZeneca, Pfizer and Moderna, asking them to join its efforts in studying the risk of blood clots together in an alliance to communicate the benefits and risks of the shot and address any concerns raised among the public.8 Both Pfizer and Moderna declined the offer, while AstraZeneca accepted.