Adam Gretton, Norfolk and Norwich University Hospital; Shidlovski / shutterstock.com
Traditional X-rays, move over—there may be a new gold standard for joint imaging to assess even the smallest changes that can signal the onset of arthritis, as reported recently in the journal Scientific Reports.1
Utilizing the combined expertise of radiologists, rheumatologists and engineers, University of Cambridge researchers developed an algorithm to monitor the joints of arthritis patients based on computed tomography (CT) scan images. The researchers used the technique, called joint space mapping (JSM), to analyze CT images of cadaver hip joints and found it could identify even the most nuanced narrowing of the space between bones. The technique, which uses color-coded images, was twice as sensitive as traditional diagnostic methods in showing small changes in joint structure.
Currently, arthritis treatment at its inception in a joint is limited, which may be due to the fact that early-stage arthritis is difficult to detect. “One of the major frustrations in the treatment of osteoarthritis is that we don’t currently have an effective cure other than a surgical joint replacement, which is costly, invasive and not without its own complications,” says lead author Tom Turmezei, MPhil, MA, BMBCh, PhD, FRCR,
formerly a clinical PhD Fellow in Cambridge University Hospital’s Department of Engineering in Cambridge, England, and now a consultant radiologist at the Norfolk and Norwich University Hospital in Norfolk, England. His research interest is in quantitative cross-modality imaging of the hip joint for biomarker development in osteoarthritis.
Dr. Turmezei and his team were interested in developing a method to help physicians get a jump on early diagnosis of osteoarthritis in the hope that early treatment may slow disease progression. Lifestyle modifications, such as weight loss, and physical therapy that strengthens muscles supporting joints can help delay arthritis progression, and pain relievers can control symptoms up to a point. However, none of these can ensure a patient’s arthritis will not progress to the point the patient requires surgery.
“Nonetheless, if we can identify those at risk of joint disease earlier, these supportive therapies stand more chance of being effective for longer,” says Dr. Turmezei, whose areas of expertise include osteoarthritis, magnetic resonance imaging, computed tomography, medical image analysis and 3D mesh processing from the university’s engineering department.
‘
If we can identify those at risk of joint disease earlier, these supportive therapies stand more chance of being effective for longer.’ —Dr. Turmezei
Dr. Turmezei, who has been involved with several cross-disciplinary musculoskeletal research projects, works with a combination of clinical CT and magnetic resonance imaging (MRI) to develop new 3D imaging analysis techniques that aim to be sensitive enough to detect the slightest disease changes in bone and cartilage.
“Another important aim of this work is to develop effective techniques that can help identify whether a novel treatment is working effectively, sooner,” Dr. Turmezei says. “An example of such a treatment being developed is regenerative cell therapy, and our groups are also supporting 3D image analysis in a clinical trial involving such a treatment.” (See more about that trial here: http://adipoa2.eu.)
For rheumatologists, this work carries three main implications. First, you will
be able to see the joint and disease-related changes in 3D with color in a way not previously possible, helping you and your patients understand what is happening more clearly. Second, with further research, the technique should help you identify earlier than before who is at risk of developing joint disease or who is developing disease rapidly, so you can start treatments sooner. And third, it should facilitate research leading to effective new treatments for joint disease.
Further research is underway to help clarify whether this tool can identify individuals at risk of joint disease when they don’t have traditional imaging features that would show they have disease. One such collaboration is underway with the Icelandic Heart Association in Reykjavik, Iceland, which is working with CT imaging of the hip joints from the AGES cohort, a population study intended to identify the role of aging, genetic and environmental factors in various common conditions.
Another collaboration is underway with Professor Neil Segal from the University of Kansas Medical Center in Kansas City, where researchers are working to establish what this tool can reveal about the knee joint and how it changes with progressing knee osteoarthritis. Professor Segal has also independently developed a 3D joint space width measurement tool.
Dr. Turmezei says of the study, “We will be looking at how we can take the best from both to improve our techniques and better understand how joints change over time with disease.”
Radiologists will still be needed to make important decisions based on imaging, just as they will with the imminent arrival of computer-aided diagnosis and artificial intelligence into regular radiological practice. The JSM technique is designed to furnish radiologists and other clinicians with an accurate and reliable view of joints, enabling them to visualize and understand the disease better, particularly when looking for small changes that might otherwise be overlooked.
Although CT has been used for several years to look at joints when planning surgical procedures, such as joint replacements, fixing fractures or checking whether surgery has been successful, Dr. Turmezei says that until recently no one had thought to look at the data in these scans in the way his team proposed. “It seems obvious in retrospect, but it took collaboration to see the potential in this and make it reality,” he says.
Improvements in CT acquisition techniques have made the prospect of joint imaging more easily justifiable because better dose reduction techniques reduce radiation levels for each examination, making it safer for patients to undergo these scans.
Dr. Turmezei says, “The doses [of radiation] involved are much less than they used to be. Certain CT acquisition techniques can involve a dose similar to a plain radiograph of the joint, for example, with a new wave of dedicated peripheral CT scanners that image just the limbs.”
As a result, Dr. Turmezei says he could imagine yearly screening of individuals at risk of developing osteoarthritis might be appropriate, particularly among individuals with a genetic predisposition, with a previous injury to a joint or who have been involved in activities that increase the joint stress, such as elite athletics. “The exact value of such scanning programs would need to be established with further research,” he says.
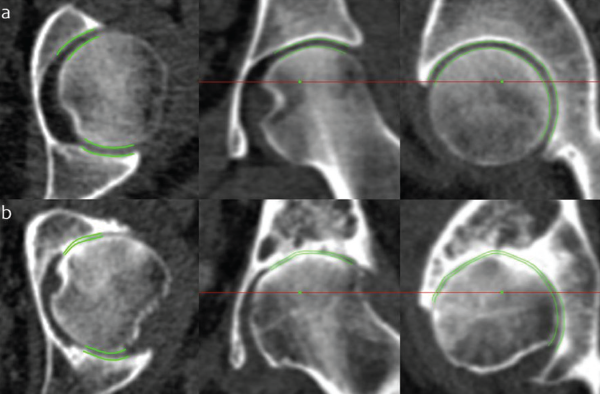
JSM performance at the extremes of disease. Joint space margins are shown in green as seen in axial, coronal and sagittal planes (left to right) at two left hips. (a) An individual with a radiographic K&L grade of 0 (no disease) and minimum joint space width of 2.0 mm. (b) An individual with a radiographic K&L grade of 3 (moderate disease) and minimum joint space width of 0 mm. This demonstrates that JSM is still able to represent joint space width as it approaches zero, important for analysis in advanced disease.
Credit: Reprinted by permission from “A new quantitative 3D approach to imaging of structural joint disease,” Scientific Reports, under the Creative Commons Attribution 4.0 International (CC BY) license (https://creativecommons.org/licenses/by/4.0/legalcode).
CT or MRI?
The researchers focused on CT scans rather than MRIs because the 3D information is richer, with better spatial resolution in all three planes. “We have been able to adapt the technique to work with MRI, but this has mainly focused on 3D mapping of cartilage thickness at the knee, which MRI is far better at visualizing,” Dr. Turmezei says. “It is still important to consider [CT] joint space mapping as complementary to other imaging techniques such as MRI, which has its own strengths over CT, such as the ability to depict soft tissues such as cartilage and ligaments as well as inflammation.” CT does better show mineralized structures, such as bone.
As MRI techniques develop, however, a similar 3D joint space mapping approach will likely become possible with clinical MRI data. Dr. Turmezei’s PhD thesis in 2017 showed it was feasible, but the sequences needed to do it are not in everyday clinical use. “One of the great strengths of CT joint space mapping is that it uses regular CT imaging data that can be acquired on any hospital scanner right now,” he says.
Prior to joining together, two of the researchers had already been collaborating for a few years, using cortical bone mapping to investigate thickness changes in the diagnosis and treatment of osteoporosis. With his background in radiology, Dr. Turmezei was able to bring a new set of clinical problems around osteoarthritis imaging for the team to solve.
Because his PhD was in the engineering department, Dr. Turmezei says he had to learn advanced math and coding to develop and validate the algorithm, and that experience brought him skills he continues to use in his research and clinical practice.
“I feel very lucky to have worked with and between these research groups that continue to be committed to solving clinical problems with novel engineering solutions,” he says. “The keystone to the success of these collaborations is the ability to identify meaningful clinical problems that need a solution. Otherwise time is wasted on developing tools that don’t have any relevant clinical utility.”
Renée Bacher is a frequent contributor to ENT Today and the AARP Bulletin. Her article about heart disease in women, “This Story Could Save Your Life,” won a recent National Health Information Award for Family Circle magazine.
Reference
- Turmezei TD, Treece GM, Gee AH, et al. A new quantitative 3D approach to imaging of structural joint disease. Scientific Reports. 2018 Jun 18;8(9280).