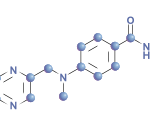
 A randomized, placebo-controlled, double-blind, six-month, multicenter Phase 3 study examined the efficacy and safety of the oral Janus kinase inhibitor, tofacitinib, in patients with psoriatic arthritis (PsA) who had an inadequate response to tumor necrosis factor-α (TNF-α) inhibitors.1
A randomized, placebo-controlled, double-blind, six-month, multicenter Phase 3 study examined the efficacy and safety of the oral Janus kinase inhibitor, tofacitinib, in patients with psoriatic arthritis (PsA) who had an inadequate response to tumor necrosis factor-α (TNF-α) inhibitors.1
Study participants had a PsA diagnosis for at least six months, with active disease at screening and baseline. Three hundred and ninety-four patients were treated and randomized into a 2:2:1:1 ratio to receive 5 mg (n=132) or 10 mg tofacitinib twice daily (n=132); or placebo to 5 mg (n=66) or 10 mg tofacitinib twice daily (n=65). Patients who were randomized to receive placebo were blindly advanced to a dosage of twice daily tofacitinib at Month 3.
Use of one conventional synthetic disease-modifying anti-rheumatic drug (DMARD) was required for study entry and continuation. The study’s primary endpoints were the change from baseline in Health Assessment Questionnaire Disability Index (ΔHAQ-DI) at Month 3 and the ACR20 response rate.
Approximately 92% of patients (n=361) completed Month 3, and approximately 88% of patients (n=345) finished Month 6. Baseline disease activity/characteristics and patient demographics were similar across all groups. The primary endpoints were statistically and significantly greater with 5 mg and 10 mg tofacitinib-treated patients compared with placebo-treated patients at Month 3, effects that persisted through Month 6.
The ACR20 response was superior for 5 mg and 10 mg tofacitinib-treated patients compared with placebo-treated patients, which was noted as early as Week 2 of the study (26.7% and 28.8% vs. 13.0%; P≤0.05). Additionally, the Psoriasis Area and Severity Index (PASI) at Month 3 was achieved by 14% of placebo-treated patients, 21% of the 5 mg tofacitinib-treated patients and 43.2% of the 10 mg tofacitinib-treated patients at Month 3, which was considered statistically significant (P<0.0001). The PASI response continued to increase through Month 6 for all treatment groups.
The serious adverse event rate was low, as were study discontinuations due to adverse events. The study had two discontinuations (3%) in the 5 mg tofacitinib-treated patient group, three discontinuations (4.6%) in the 10 mg tofacitinib-treated group, five discontinuations (3.8%) in the placebo to 5 mg tofacitinib-treated group, and 11 discontinuations (8.3%) in the placebo to 10 mg tofacitinib-treated group. The most common adverse events were headache (4.5–9.1%), nasopharyngitis (1.5–10.7%) and upper respiratory tract infection (5.3–10.8%). No deaths or malignancies were reported in this short-term study, and no new safety risks were identified.
Michele B. Kaufman, PharmD, BCGP, is a freelance medical writer based in New York City and a pharmacist at New York Presbyterian Lower Manhattan Hospital.