Plaquenil is a mainstay of treatment for inflammatory arthritis and skin symptoms.
Image Credit: Alice Day/shutterstock.com
In 1984, I wrote my first prescription for the antimalarial drug, hydroxychloroquine (Plaquenil), for a 28-year-old woman with SLE. She was considerably overweight, with inflammatory arthritis and a photosensitive rash, and I worried that oral corticosteroids would tip her over into diabetes. I presented the case to my attending, Steven Malawista, MD, at the Yale New Haven Medical Center (Conn.) where I was a visiting internal medicine resident, and to my relief, he agreed with my plan.
But at that time, in reviewing potential side effects, I had to scare the “bajeezus” out of my patient: “Plaquenil is usually very safe,” I told her, “but it can damage your retina, and we need to obtain a baseline eye exam and then monitor your eyes every six months for as long as you are on the drug.”
I can still remember her pointing an accusing finger at my nose and saying, “You mean to tell me that because I have this ugly rash and some aches and pains, you’re going to give me a medicine that might make me go blind?”
“Well, ugh, no, I mean we need to be cautious. Just a minute. Let me get my attending, and he can talk to you.”
Luckily, Dr. Malawista was able to convince our patient to take the prescription. After three months, it worked; her rash and inflammatory arthritis resolved. She didn’t go blind. Actually, over the ensuing 31 years, none of my patients have suffered visual loss on Plaquenil. That is a wonderful story in itself: How many of our medications have stood the test of time as effective medications and are safer than originally believed? And how did we make the leap from treatment of malaria to the recognition of Plaquenil’s value in the management of lupus and rheumatoid arthritis?
In the treatment of SLE, [Plaquenil is] widely regarded as a cornerstone drug in the management of skin & arthritis symptoms.
History of Antimalarials
The history of antimalarials dates back to the 1630s in Peru, where, in all likelihood, malaria was not endemic, and an extract from the “fever tree,” which grew on the eastern slopes of the Andes. Antonio de Calancha, an Augustinian monk, was the European first credited with describing the medicinal effects of the fever tree, “whose bark, of the color of cinnamon, made into powder amounting to the weight of two small silver coins and given as a beverage, cures the fevers and tertiana; it has produced miraculous results in Lima.”1
de Calancha may have been referring to the wife of the viceroy of Peru, Countess Anna del Chinchon, who miraculously recovered from a febrile illness when given an extract from the bark obtained from native healers. Unknown at the time, the bark contained a number of alkaloids with surprising and enduring pharmacologic properties, including the antimalarial compound, quinine, and the antiarrhythmic, quinidine.
The bark soon made its way to Europe, where, over time, its success in the treatment of ague (i.e., swamp fever or malaria) became well established. In 1742, the physician/taxonomist, Carl Linnaeus, classified the “fever tree” Cinchona officialis, after the legend of Countess Anna del Chinchon.2
In 1820, chemists Pierre Joseph Pelletier and Joseph Canentou isolated and purified quinine from Cinchona bark.3 From that time on, a standardized dose and purity for the drug was established. Eventually, the Dutch developed industrial-sized plantations of Cinchona in Java to supply the world antimalarial market.
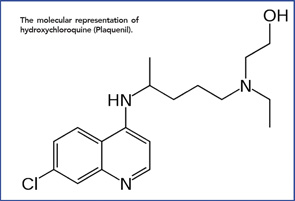
The molecular representation of hydroxychloroquine (Plaquenil).
When Japan overran Java in World War II, the world supply of natural quinine shrunk dramatically, just as Allied troops were pouring into the South Pacific. The solution? A synthetic version of quinine, quinacrine (Atabrine), became the drug of choice for the prevention of malaria.4 And the drug worked. As many as 3 million Allied soldiers ingested quinacrine prophylaxis daily for up to three years, with only rare side effects.5
From Malaria to Lupus & RA
Anecdotal reports describing improvement in lupus and rheumatoid arthritis (RA) by Allied soldiers on long-term quinacrine culminated in the first published report by Page in the Lancet, in 1951.6 Following this, other papers followed, and the drugs became a mainstay in the treatment of SLE and increasingly prescribed in RA. Over time, other quinine derivatives were introduced by the FDA: chloroquine (Aralen) in 1953 and hydroxychloroquine (Plaquenil)—differing from chloroquine by a single hydroxyl group side chain, in 1955.
There were missteps. If quinine derivatives were effective in immune-mediated disease, why not combine three antimalarial drugs in a single pill? Triquen (containing 25 mg chloroquine, 50 mg hydroxychloroquine and 65 mg quinacrine) was widely prescribed—until it was withdrawn from the market in 1972 due to frequent retinal toxicity. The chief culprit, chloroquine, became a drug of last resort for refractory SLE rashes. Quinacrine was widely viewed as a safer, but perhaps less effective, drug and eventually could be obtained only through compounding pharmacies (although Edmund Dubois, MD, and later, Daniel J. Wallace, MD, at UCLA, continued to prescribe it regularly for their lupus patients).7
That left Plaquenil. In the treatment of SLE, it’s widely regarded as a cornerstone drug in the management of skin and arthritis symptoms.
In RA, Plaquenil is prescribed as monotherapy in mild disease or combined with other disease-modifying medications to control more aggressive disease.
Plaquenil is included in the World Health Organization’s List of Essential Medicines, a listing of the essential medications needed in a basic health system.8 The drug, in its generic form, is cheap and generally well tolerated. It does not require regular laboratory monitoring. It can be continued in patients receiving antibiotics for bacterial infections. Although it is considered to be a category C drug, it is generally regarded as safe for women to continue on during pregnancy, and lactating women may breastfeed while taking the drug.9
The Research Says …
Like the Cinchona tree, Plaquenil has a number of additional valuable and surprising properties.
The beneficial effects of Plaquenil on lipid profiles have been confirmed in numerous trials.10-12 These effects are relatively small, with decreased LDL and triglycerides, but in RA and SLE, any medication that can partially mitigate the unfavorable lipid profiles of patients often on corticosteroids is not insignificant.
In 1990, the Annals of Internal Medicine published an intriguing article by Antonio Quatraro, MD, “Hydroxychloroquine in Decompensated, Treatment-Refractory Noninsulin-Dependent Diabetes Mellitus: A New Job for an Old Drug?”13 This small, prospective, randomized, placebo-controlled trial found clinically meaningful improvement in glucose control with Plaquenil.
Other studies followed. Mary Chester M. Wasko, MD, at the University of Pittsburgh, determined in a large prospective trial of 4,905 adult RA patients that long-term use of Plaquenil reduced the risk of diabetes mellitus.14 Risk reduction was both dose and duration dependent; RA patients taking 400 mg Plaquenil daily for more than than four years had a 77% lower risk of developing diabetes than RA patients who never received the drug.
Lastly, Rekedal and colleagues found significant reductions in glycosylated hemoglobin in established diabetic patients with rheumatic disease after initiation of Plaquenil compared with methotrexate.15 The mean change in HbA1c of 0.66 from baseline was only slightly less than with frequently prescribed oral hypoglycemics.15
For patients at risk of thrombosis, Plaquenil may lower this risk. Michelle Petri, MD, found that in her cohort of 2,000 SLE patients at Johns Hopkins, those with phospholipid/lupus anticoagulants on long-term Plaquenil had a lower risk of thrombosis than those who never took the drug.16 Studies in the early 1970s explored the antithrombotic effects of Plaquenil in patients undergoing hip arthroplasty.17 Although most studies demonstrated moderate efficacy in preventing postoperative deep venous thrombosis, newer regimens with heparin and warfarin have proved superior.
Additional Potential Uses
A number of additional disorders exist for which a trial of Plaquenil is more than reasonable. Dermatologists use the drug for polymorphous light eruptions, granuloma annulare, porphyria cutanea tarda and necrobiosis lipoidica diabeticorum. Rheumatologists are often pleasantly surprised at follow-up that their patient’s undifferentiated connective tissue disorder (UCTD), palindromic rheumatism or inflammatory osteoarthritis has improved. A subset of hypocomplementemic urticarial vasculitis may respond to Plaquenil. Current EULAR recommendations suggest that Plaquenil be considered as an adjunctive treatment in chronic (but not acute) calcium pyrophosphate deposition (CPPD) arthropathy.18
The drug is particularly well suited for a subset of patients. How often have we seen an unfortunate young adult with active RA, who, prior to consultation, has tried every supplement and antiinflammatory diet, but then declines a trial of methotrexate or a biologic out of fear of side effects? Even after explaining that serious side effects with these more powerful drugs are uncommon and that the benefits far outweigh the risks, I’m often up against a wall of distrust and fear. Rather than have the patient leave the office empty-handed, I’m usually able to convince them to take a prescription for Plaquenil. It keeps them in the fold. It establishes trust. Over time, I can usually, if necessary, add a more effective drug.
Cautions & Dosing
Of course, no drug is absolutely safe. Plaquenil rashes are seen relatively frequently. Some patients simply don’t tolerate the drug. Diarrhea and headaches may be a reason for discontinuation, and rare cases of neuromyopathy have been reported. A 45-year-old patient of mine on Plaquenil for 12 years developed a cardiomyopathy. An endomyocardial biopsy was consistent with Plaquenil toxicity. The drug was stopped, but her ejection fraction never recovered.
We need to remain vigilant regarding retinal toxicity by dosing the drug based on lean body weight and remembering that most reported cases of retinal toxicity have been in patients prescribed greater than 5.5 mg/kg/day. (Or alternatively, as Ronald Melles, MD, and Michael Marmor, MD, recently suggested, dosage should be no greater than 5.0 mg/kg real weight).19 Because Plaquenil is primarily cleared by the kidneys, it’s prudent to decrease the dose in patients with renal insufficiency.
Our patients require Plaquenil eye check-ups, but these recommendations have evolved from earlier years in which a baseline assessment was followed indefinitely with check-ups every six months to the current recommendations by the American College of Ophthalmology that after a baseline evaluation, a second eye exam is not required for five years unless there are unusual risk factors.20
Improved Cancer Survival?
Lastly, who would have predicted that Plaquenil may play a role in the treatment of malignancy? Autophagy is the process of sequestration of stressed or damaged organelles and proteins into lysosomes where they are subsequently degraded. In established malignancy, an “autophagy switch” occurs, with increased autophagy activity and decreased apoptosis, both of which favor malignant cell survival. Plaquenil, through pH dependent inhibition of lysosomal activity, may partially block cancer upregulation of autophagy.21
To date, more than 30 current trials are underway to assess whether the addition of Plaquenil to standard chemotherapy improves cancer survival.
Improved Lives
Taken together, the portfolio of Plaquenil is impressive. We are indebted to the “fever tree,” the Cinchona tree of South America, for quinine and the subsequent development of Plaquenil. Medical science has given us a medication that improves the quality of our patients’ lives, year after year after year. Let us praise Plaquenil.
Charles Radis, DO, is a rheumatologist in Portland, Maine, and director of clinical research for Rheumatology Associates.
Acknowledgment
The author thanks Mary Chester M. Wasko, MD, MSc, director of the Rheumatology Division, West Penn Hospital, Allegheny Health Network, for her review assistance.
Editor’s note: The use of the brand name, Plaquenil, does not signify an endorsement of the product by the ACR or The Rheumatologist.
References
- Duran-Reynals ML. The Fever Bark Tree: The Pageant of Quinine. Doubleday. 1946. 24.
- Jaramillo-Arango JA. Critical review of the basic facts in the history of cinchona. Journal of the Linnean Society (Botany). 1949 Mar;53(352):272–311.
- Delepine M. Joseph Pelletier and Joseph Caventou. Chem Educ. 1951;28(9):45.
- Office of the Surgeon General: Circular letter No. 153: The drug treatment of malaria, suppressive and clinical. JAMA. 1943;123:205–208.
- Goodman LS, Gilman A: The Pharmacological Basis of Therapeutics (ed 2). New York, Macmillan, 1954;1167–1173.
- Page F. Treatment of lupus erythematosus with mecaprine. 1951. Lancet. 2:755–758.
- Wallace DJ. The use of quinacrine (atabrine) in rheumatic diseases: A reexamination. Seminars in Arthritis and Rheumatism. 1989 May;18(4):282–297.
- WHO Model List of Essential Medicines. October 2013. World Health Organization.
- Motta M, Tincani A, Faden D, et al. Follow-up of infants exposed to hydroxychloroquine given to mothers during pregnancy and lactation. J Perinatol. 2005 Feb;25(2):86–89
- Rahman P, Gladman DD, Urowitz MB, et al. The cholesterol lowering effect of antimalarial drugs is enhanced in patients with lupus taking corticosteroid drugs. J Rheumatol. 1999 Feb;26(2):325–330.
- Morris SJ, Wasko MC, Antohe JL, et al. Hydroxychloroquine use associated with improvement in lipid profiles in rheumatoid arthritis patients. Arthritis Care Res (Hoboken). 2011 Apr;63(4):530–534.
- Kerr G, Aujero M, Richards J, et al. Associations of hydroxychloroquine use with lipid profiles in rheumatoid arthritis: Pharmacologic implications. Arthritis Care and Research. 2014 Nov;66(11):1619–1626.
- Quatraro A, Consoli G, Magno M, et al. Hydroxychloroquine in decompensated, treatment-refractory noninsulin-dependent diabetes mellitus: A new job for an old drug? Annals of Internal Medicine. 1990 May 1;112(9):678–681.
- Wasko MC, Hubert HB, Lingala VB, et al. Hydroxychloroquine and risk of diabetes in patients with rheumatoid arthritis. JAMA. 2007 Jul 11;298(2):187–193.
- Rekedal LR, Massarotti E, Garg R, et al. Changes in glycosylated hemoglobin after initiation of hydroxychloroquine or methotrexate treatment in diabetes patients with rheumatic diseases. Arthritis Rheum. 2010 Dec;62(12):3569–3573.
- Fangtham M, Petri M. 2013 update: Hopkins lupus cohort. Curr Rheumatolol Rep. 2013 Sep;15(9):360.
- Wu TK, Tsapogas MG, Jordan FR. Prophylaxis of deep venous thrombosis by hydroxychloroquine sulfate and heparin. Surg Gynecol Obstet. 1977 Nov;145(5):714–718.
- Zhang W, et al. EULAR recommendations for calcium pyrophosphate deposition. Part II: Management. Annal Rheum Dis. 2011 Apr;70(4):571–575.
- Marmor MF, Kellner U, Lai TY, et al. Revised recommendations on screening for chloroquine and hydroxychloroquine retinopathy. Ophthalmology. 2011 Feb;118(2):415–422.
- Melles RB, Marmor MF. The risk of toxic retinopathy in patients on long-term hydroxychloroquine. JAMA Ophthalmol. 2014 Dec;132(12):1453–1460.
- Sui X, Chen R, et al. Autophagy and chemotherapy resistance: A promising therapeutic target for cancer treatment. Cell Death Dis. 2013 Oct 10;4:e838.