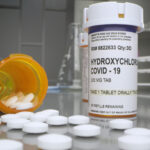
ZURICH (Reuters)—The World Health Organization (WHO) on Tuesday promised a swift review of data on hydroxychloroquine (HCQ), probably by mid-June, after safety concerns prompted the group to suspend the malaria drug’s use in a trial on COVID-19 patients.
U.S. President Donald Trump and others have pushed HCQ as a possible coronavirus treatment, but the WHO on Monday called time in its multi-country trial, called Solidarity.
A study in British medical journal The Lancet found patients getting HCQ had increased death rates and irregular heartbeats, prompting the WHO’s intervention.
“A final decision on the harm, benefit or lack of benefit of HCQ will be made once the evidence has been reviewed by the Data Safety Monitoring Board,” the body said in a statement. “It is expected by mid-June.”
Those already in its by-now, 17-country study of thousands of patients who have started HCQ can finish their treatment, the WHO said.
Newly enrolled patients will receive other treatments being evaluated in Solidarity, including Gilead Science’s remdesivir and AbbVie’s Kaletra/Aluvia.
Separate HCQ trials, including a 440-patient U.S. study by Swiss drug maker Novartis, are continuing even while the WHO slows down. Novartis and rival Sanofi have pledged to donate tens of millions of doses of the drug, also used in rheumatoid arthritis and lupus, if it is shown to be effective and safe for COVID-19.
Novartis said The Lancet study, although covering 100,000 people, was only “observational” and not capable of demonstrating a causal link between HCQ and side effects.
“We need randomized, controlled clinical trials to clearly understand efficacy and safety,” a Novartis spokesman said.