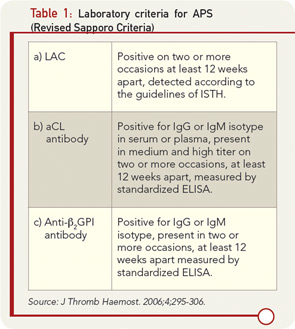
Although international consensus guidelines for the determination of LAC have been published and revised, “standardized” tests for detection of aCL and anti-β2GPI have remained elusive. Furthermore, despite more than 7,000 publications related to the clinical use of aPL antibody tests, a consensus on clinical recommendations has been difficult to achieve. This difficulty appears related to suboptimal design in clinical studies and lack of laboratory standardization in areas such as: 1) units of measurement; 2) calibration curves; 3) determination of cut-off values; and 4) laboratories not performing the tests according to established guidelines. Significant interassay and interlaboratory variation in the results of both aCL and anti-β2GPI testing still exists, affecting the consistency of the diagnosis of APS.8
Over the years, international workshops have labored to standardize the laboratory test in this area. These workshops include the APL European Forum, the Australasian Anticardiolipin Working Party, the College of American Pathologists, the National External Quality Assessment Scheme, and the Standardization Subcommittee on Lupus Anticoagulant and Phospholipid-Dependent Antibodies of the International Society of Thrombosis and Hemostasis (ISTH). Although some laboratories can obtain reliable testing results, there is still wide interlaboratory variation despite efforts at standardization. This situation may result from laboratories performing aPL antibody assays with their own protocols or using commercial kits that do not conform to the proposed guidelines for these tests. Standardization of tests or reevaluation of standardization is important because APS is related to serious complications such as thrombosis and pregnancy loss; missing a diagnosis because of laboratory variability could have serious medical consequences. The use of semi- or fully automated analyzers and commercial kits instead of in-house assays poses additional challenges to the process of standardization.8
Criteria aPL Task Force and Work Recommendations
To address the challenges on aPL antibody testing described above, an international criteria aPL task force of researchers and scientific leaders in the field was formed prior to the 13th International Congress on Antiphospholipid Antibodies in Galveston, Texas, in April 2010 (APLA 2010). The task force was further divided into three subgroups that were charged by the congress chair to address, in an evidence-based manner, various topics related to the testing of aCL, anti-β2GPI, and LAC.
To accomplish its mission, the task force considered published information, the results of a survey distributed among congress attendees, and discussions that occurred during a special preconference workshop. On the basis of this approach, the task force reached several conclusions and proposed recommendations discussed here and summarized in Table 2; this information was recently published.9
Subgroup 1
Emerging new detection technologies and automated platforms with constantly evolving laboratory environments have made it necessary to update the standardization process and to revise existing protocols. Moreover, at present, there are no international consensus guidelines for aCL and anti-β2GPI. The task force members prepared a manuscript that included international guidelines centering on the characteristics of immunoassays for the measurement of aCL and anti-β2GPI antibodies.10 These guidelines are for the use of both consumers and manufacturers. The topics covered by guidelines include preanalytical issues (type of specimen), analytical issues (type and source of the antigen, method of calibration and quantitation, precision of the measurements, the issue of single or duplicate testing, interference testing, cut-off calculation, and expression of the result), and postanalytical issues (method of reporting results and interpretative comments).