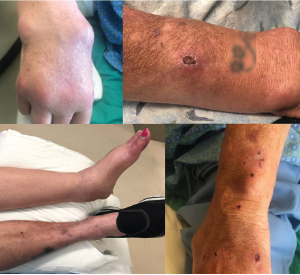
Figure 1: Salient Features of the Patient Exam
The top left panel shows synovitis in the right second through fourth metacarpal phalangeal joints, with large subcutaneous cysts involving the distal forearm and wrist. The top right panel shows a right forearm lesion with overlying ulceration. The bottom left panel demonstrates marked swelling in left ankle. The bottom right panel shows scattered ulcerations on the upper left arm.
A 61-year-old Caucasian woman with a history of seropositive rheumatoid arthritis (RA) was hospitalized for a several-month history of progressively worsening left ankle pain and swelling.
She had been unable to bear weight on her left leg for several days and did not notice improvement in symptoms with 20 mg of prednisone daily, which she had increased to 40 mg daily a few days prior to admission. Her right wrist and hand were also painful and swollen. Over the preceding months, she had noted progressively enlarging, painful nodules on her right forearm, one of which had spontaneously drained yellow-tinged fluid. She had also developed ulcerations over some of the nodules and the contralateral forearm. She did not have any preceding trauma to the right upper extremity. A review of systems was notable for several months of intermittent night sweats.
She had several pug dogs at home; one had recently died, and both had been experiencing rashes and skin issues. She reported no other pets, nor any unusual personal or occupational exposures.
The patient had been diagnosed with seropositive RA at age 55 and had been previously treated with multiple biologic and conventional synthetic disease-modifying anti-rheumatic drugs (DMARDs), most recently with good therapeutic response to rituximab.
Unfortunately, she had lost her private insurance coverage, resulting in poor access to care and being off all biologics for a year at the time of her presentation. However, she reported taking 20 mg of prednisone daily for the preceding four or five months.
Other pertinent medical history included a previous left ankle infection, which, per the patient, had been treated successfully with surgical washout and intravenous antibiotics the prior year.
After an initial work-up for an infectious etiology proved negative, a rheumatologist was consulted for treatment recommendations for a presumptive inflammatory arthritis flare with concern for rheumatoid nodules or, possibly, ganglion cysts involving the right upper extremity.
Further history and review of outside records confirmed the history of polymicrobial left ankle septic arthritis, with cultures at the time yielding coagulase-negative Staphylococcus, Corynebacterium and Sporothrix schenckii. Outside records indicated she had completed her recommended course of antibiotics for the bacterial infections; however, her insurance had lapsed prior to completion of the recommended six-month course of antifungal treatment.
On exam, the patient was Cushingoid and chronically ill appearing, with normal vital signs. Her musculoskeletal exam was notable for synovitis in her right second through fifth metacarpophalangeal joints, as well as in her right wrist, right elbow and left ankle (see Figure 1). Her left ankle was warm and tender, with pain on both active and passive range of motion. She was also noted to have several tender, fluctuant, cystic lesions on the right forearm, as well as shallow ulcerations on both upper arms (see Figure 1).