The COVID-19 pandemic has filled hospitals with patients with rapidly progressive respiratory failure and diffuse bilateral opacities on chest X-ray.1 Additionally, many patients with severe COVID-19 develop acute kidney injury and require dialysis.2 Pulmonary-renal syndromes are also important to consider in this setting. Although alveolar hemorrhage is a cardinal feature of this syndrome, many patients do not present with gross hemoptysis.3
We present a case of diffuse alveolar hemorrhage (DAH) and acute renal failure secondary to microscopic polyangiitis (MPA).
Case Description
A 38-year-old Hispanic man with past medical history of asthma presented to the emergency department in June 2020 with progressive shortness of breath over two weeks; this was accompanied by a loss of taste and smell, as well as poor appetite. He also reported associated nausea, vomiting and diarrhea. Upon presentation, he was tachypneic and in respiratory distress. He was hypoxic, with oxygen saturation 85% on 5 L/min. of oxygen by nasal cannula.
His initial laboratory results were significant for hemoglobin of 6.0 g/dL (reference range [RR]: 13.5–18.0 g/dL); hematocrit of 18.5% (RR: 40–54%); platelets 161,000/μL (RR: 150,000–450,000/μL); potassium of 7.3 mmol/L (RR: 3.5–5.1 mmol/L); bicarbonate of 11 mmol/L (RR: 24–32 mmol/L); creatinine of 30.7 mg/dL (RR: 0.7–1.3 mg/dL); blood urea nitrogen (BUN) of 153 mg/dL (RR: 9–20 mg/dL); a urinalysis showing cloudy urine with >180 red blood cells/high-powered field (hpf; RR: 0–3/hpf); proteinuria of 100 mg/dL (RR: 0–14 mg/dL);
and a white blood cell count of 14/hpf (RR: 0–4/hpf).
Arterial blood gas demonstrated a pH of 7.249 (RR: 7.35–7.45); pCO2 of 25.1 mmHg (RR: 35–45 mmHg); pO2; 63 mmHg (RR: 80–105 mmHg); and bicarbonate 11.0 mmol/L (RR: 24–32 mmol/L). Amylase and lipase were mildly elevated to 113 U/L (RR: 29–103 U/L) and 114 U/L (RR: 11–82 U/L), respectively. White blood cell count, blood glucose, aspartate transaminase (AST), alanine transaminase (ALT), bilirubin, alkaline phosphatase, lactate dehydrogenase (LDH), and troponin were all within normal limits.
A nasopharyngeal swab was performed and tested for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) RNA, which was not detected.
A chest X-ray on admission showed bilateral airspace opacities (see Figure 1), and a chest computed tomography (CT) scan showed extensive diffuse groundglass opacities and cystic changes in both the upper and lower lobes of the lungs (see Figure 2).
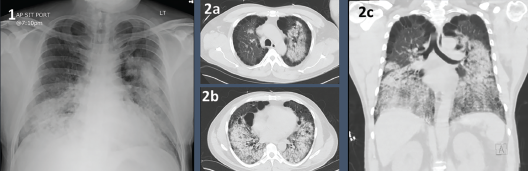
Figures 1 & 2. Figure 1: Chest X-ray with bilateral airway opacities.
Figure 2A–C: Chest CT showing extensive bilateral upper and lower lobe airway disease.
The patient was admitted to the intermediate level of care but rapidly declined, requiring transfer to the intensive care unit (ICU). At that point, despite the initial negative test, the working diagnosis was of severe COVID-19 pneumonia due to the epidemiological considerations, his initial presentation with loss of taste and smell, and respiratory failure with bilateral pulmonary infiltrates.
In the ICU, he went on to develop severe acute respiratory distress syndrome (ARDS) and required intubation. Following intubation, he was noted to have a substantial quantity of bloody secretions in the endotracheal tube and underwent bronchoscopy, which showed significant blood in the airways. This blood did not clear with sequential lavage.
For his acute renal failure, continuous venovenous hemofiltration was started. Given the patient’s persistent bloody secretions, pulmonary-renal syndrome due to underlying vasculitis was suspected. He was started on plasmapheresis and pulse-dose glucocorticoids with 1 g methylprednisolone daily for five days.
He remained in the ICU for a total of 12 days. HIs ICU course was further complicated by worsening acute blood loss anemia and associated coagulopathy, requiring multiple transfusions with packed red blood cells, platelets, fresh frozen plasma and cryoprecipitate. He had refractory hypoxemia, requiring high-fraction inspired oxygen (FiO2) up to 80–90%, as well as inhaled nitric oxide.
An evaluation for rheumatic disease was pursued (see Table 1). His serologies were positive for perinuclear anti-neutrophil cytoplasmic antibody (P-ANCA) at a titer of 1:320 and anti-myeloperoxidase (MPO) antibodies at 41 units. His complement levels were low: C3 of 43 mg/dL and C4 of 13 mg/dL. Of note, cytoplasmic (C) ANCA, anti-nuclear antibody, anti-cardiolipin antibody, lupus anticoagulant, double-stranded DNA antibody and anti-glomerular basement membrane antibody tests were negative. A second nasopharyngeal swab was performed on the second day of hospitalization, which again did not detect SARS-CoV-2 RNA.
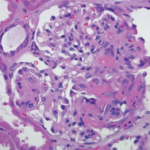
The patient received a total of seven daily sessions of plasmapheresis. Given worsening renal failure and ARDS, a renal biopsy was performed on hospital day 7, which revealed pauci-immune glomerulonephritis with crescent formation in 9 of 10 non-sclerotic glomeruli, consistent with microscopic polyangiitis with crescentic glomerulonephritis (see Figure 3).
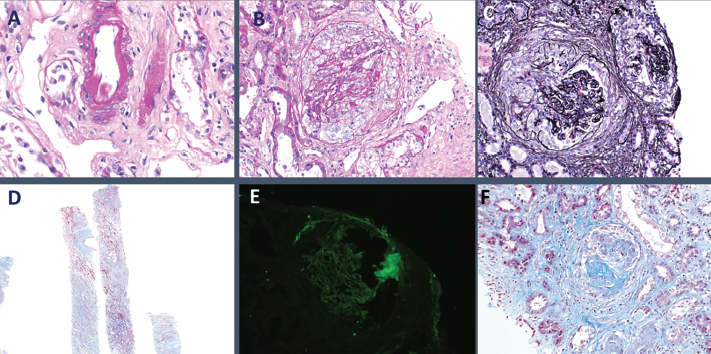
Figure 3: Histopathology from kidney biopsy. A) Arteriolar hyalinosis; B) Cellular crescents; C) Diffuse crescentic glomerulonephritis; D) Low-power objective; E) IgG negative; F) Segmental glomerulosclerosis.
After eight days of continuous venovenous hemofiltration, he transitioned to scheduled hemodialysis. He intermittently received broad-spectrum antibiotics for a total of five days due to concern for occult infection; however, all blood and respiratory cultures ultimately showed no growth.
In view of his lack of improvement clinically and no evidence of infections, on hospital day 11, he was started on 375 mg/m2 of intravenous rituximab weekly for total of four doses. Respiratory status improved remarkably with immunosuppression and plasmapheresis, and he was successfully extubated to high-flow nasal cannula the following day.
He transitioned to scheduled dialysis three times weekly, and he was ultimately discharged in good condition. He was discharged on 60 mg of prednisone daily, 1.5 g of atovaquone daily for Pneumocystis jirovecii prophylaxis and scheduled for weekly rituximab infusions to complete a four-week induction.
Since discharge, he has continued with maintenance rituximab infusions and is currently taking 15 mg prednisone daily. He remains dialysis dependent and has transitioned to home peritoneal dialysis.
Discussion
The COVID-19 pandemic is thought to have begun in Wuhan, China, in December 2019, and has developed into a pandemic of proportions not seen in modern history.1 In this setting of recurrent cases of similar clinical presentation of severe COVID-19, it is important to avoid cognitive errors that can lead to missing diagnoses with similar presentations. It is crucial to consider other causes for concurrent respiratory and renal failure.
An important cognitive error applicable to the current scenario is premature closure, which consists of believing enough information has been obtained to close the diagnosis without looking for further differential diagnoses.
A common bias that leads to premature closure is the anchoring bias, in which a diagnosis is not reconsidered despite new evidence that may undermine the initial assumption.4 An example of that applied to this case would be fixating on the fact that our patient presented during the height of the COVID-19 pandemic with respiratory symptoms, loss of taste and smell, and bilateral pulmonary infiltrates, without noting the severe renal failure with an active urinary sediment or the bloody secretions in the endotracheal tube after intubation. This is one of the most important lessons we learned from this case.
Treatment for the different types of ANCA-associated vasculitis is similar & depends on the presence of organ involvement & the level of disease activity.
The differential diagnosis for DAH is broad and includes many different pathophysiologic processes, including pulmonary capillaritis, bland hemorrhage and diffuse alveolar damage.3 The presence of DAH with glomerulonephritis, which is characteristic of pulmonary-renal, helps narrow the differential diagnosis.
Common etiologies of pulmonary-renal syndrome include microscopic polyangiitis, granulomatosis with polyangiitis (GPA), eosinophilic granulomatosis with polyangiitis (EGPA), Goodpasture syndrome and systemic lupus erythematosus (SLE). This patient was diagnosed with MPA based on the clinical presentation of alveolar hemorrhage with renal failure and hematuria, positive P-ANCA and MPO, and kidney biopsy showing pauci-immune crescentic glomerulonephritis without granulomas.5-7
MPA is an autoimmune medium- to small-vessel vasculitis grouped under ANCA-associated vasculitides (AAV). Common presentations include fever, fatigue, weight loss, arthralgias, cough, dyspnea, active urinary sediment with or without renal failure, purpura and neurologic dysfunction. Prodromal symptoms may last weeks to months without evidence of specific organ involvement.6,8,9
The diagnosis is based on clinical presentation and biopsy showing necrotizing vasculitis without immune deposits or granulomas.10,11 The presence of P-ANCA is suggestive of and helps differentiate MPA from GPA, which classically presents with C-ANCA and involves the nasopharynx.12 EGPA also presents with P-ANCA; however, it classically spares the kidneys and may involve the heart. Additionally, it is typically associated with peripheral blood eosinophilia. The presence of ANCAs against MPO is suggestive of MPA, and more specific than P-ANCA alone.9
In this case, the presence of a pulmonary-renal syndrome with active urinary sediment, elevated P-ANCA and anti-MPO antibodies led to a strong clinical suspicion for AAV, especially MPA, which prompted the kidney biopsy.
Table 1: Serology Results
| Rheumatology evaluation | Reference Range | |
|---|---|---|
| Anti-nuclear Antibody | NEGATIVE | negative |
| Myeloperoxidase Antibody | 41 (H) | |
| Proteinase 3 Ab | 3 | |
| P-ANCA | 1:320 (A) | |
| C-ANCA | <1:20 | |
| Anti-cardiolipin IgA | <9.0 | |
| DNA(DS) Ab | NEGATIVE | negative |
| JO 1 Ab, IgG | 2 | |
| RHEUMATOID FACTOR | <20 | latest RR: <30 IU/mL |
| RNP ANTIBODY | 2 | |
| SSA Antibody | 2 | |
| SSB Antibody | 4 | |
| SM Ab, IgG | 4 | |
| SCL 70 Ab, IgG | 3 | |
| JO 1 Ab, IgG | 2 | |
| Kappa/Lambda FLC Ratio | 0.96 | |
| SCL 70 Ab, IgG | 3 | |
| SM Ab, IgG | 4 | |
| Cardiolipin Ab (IgG) | <9.0 | |
| Cardiolipin Ab (IgM) | 9.7 | |
| Cryoglobulin Screen | NEGATIVE | |
| Free Kappa Light chains | 12.08 (H) | |
| Free Lambda Light chains | 12.57 (H) | |
| ADAMTS 13 Activity | 27 (LL) | |
| ADAMTS 13 Inhibitor | <0.4 | |
| GLOMERULAR BASEMENT MEMBRANE | 2 |
Abbreviations not listed in the text: Jo-1 antibody (Ab): anti-histidyl-tRNA synthetase; RNP: ribonucleoprotein; SSA, SSB: anti-Sjögren’s-syndrome-related antigens A and B; SM: Anti-Smith antibody; Scl-70 Ab: Anti-topoisomerase-1 antibodies; ADAMTS-13: ADAM metallopeptidase with thrombospondin type 1 Motif 13
Serological tests are suggestive of a diagnosis; however, guidelines recommend biopsy to confirm the diagnosis of AAV due to its treatment implications.13 The ANCA-associated small vessel vasculitis syndromes are referred to as pauci-immune because they show little to no glomerular staining for immunoglobulins or complements on immunofluorescence microscopy. Necrotizing and crescenteric glomerulonephritis are seen on light microscopy, and electron microscopy will show subendothelial edema, microthrombosis and degranulation of neutrophils without the presence of immune deposits.10,11
MPA is characterized by non‑granulomatous vasculitis with P-ANCA staining to MPO (~60%). GPA is characterized by necrotizing granulomas with adjacent necrotizing vasculitis. It more commonly presents with C-ANCA to proteinase-3 (~60%), although P-ANCA to MPO can be seen as well (~20%).9,12
Unlike the pauci-immune pattern of GPA and MPA, Goodpasture syndrome presents with a linear immunofluorescent pattern with anti-basement membrane antibodies. SLE nephritis typically presents with mesangial and subendothelial immune deposits.3 As mentioned above, EGPA typically spares the kidney, but when compromised also leads to the formation of granulomas.
Treatment for the different types of AAV is similar and depends on the presence of organ involvement and the level of disease activity. In the setting of life-threatening organ dysfunction, immunosuppression with pulse dose glucocorticoids and induction with rituximab or cyclophosphamide is recommended.13,14
Our patient received a course of pulse dose glucocorticoids and rituximab due to the more favorable safety profile when compared to cyclophosphamide. Selected critically ill patients, such as patients with rapidly progressing glomerulonephritis or diffuse alveolar hemorrhage, could be treated with plasmapheresis in the critical care setting, as we opted to do, although a recent randomized clinical trial did not support this.13,15
Patients, such as ours, often present when they are critically ill, and treatment with immunosuppression should not be delayed. If underlying AAV is a significant concern, treatment should commence immediately, with subsequent biopsy confirmation when possible.13
 Benjamin Aronow, MD, is an internal medicine intern at the University of Connecticut, Farmington, who will soon start his anesthesiology career at Boston University.
Benjamin Aronow, MD, is an internal medicine intern at the University of Connecticut, Farmington, who will soon start his anesthesiology career at Boston University.
 Eduardo Mantovani Cardoso, MD, is an internal medicine resident at the University of Connecticut, Farmington, and will soon be a rheumatology fellow at Beth Israel Deaconess Medical Center, Boston.
Eduardo Mantovani Cardoso, MD, is an internal medicine resident at the University of Connecticut, Farmington, and will soon be a rheumatology fellow at Beth Israel Deaconess Medical Center, Boston.
 Steffi Thomas, DO, is a rheumatology fellow at the University of Connecticut, Farmington.
Steffi Thomas, DO, is a rheumatology fellow at the University of Connecticut, Farmington.
 Prashant Grover, MD, is an assistant professor of medicine at the University of Connecticut, Farmington, and director of the intensive care unit at Saint Francis Hospital, Hartford, Conn.
Prashant Grover, MD, is an assistant professor of medicine at the University of Connecticut, Farmington, and director of the intensive care unit at Saint Francis Hospital, Hartford, Conn.
 Weishali Joshi, MD, is a rheumatologist at Saint Francis Hospital, Hartford, Conn.
Weishali Joshi, MD, is a rheumatologist at Saint Francis Hospital, Hartford, Conn.
References
- Wiersinga WJ, Rhodes A, Cheng AC, et al. Pathophysiology, transmission, diagnosis, and treatment of coronavirus disease 2019 (COVID-19): A review. JAMA. 2020 Aug 25;324(8):782–793.
- Fabrizi F, Alfieri CM, Cerutti R, et al. COVID-19 and acute kidney injury: A systematic review and meta-analysis. Pathogens. 2020 Dec 15;9(12):1052.
- West SC, Arulkumaran N, Ind PW, Pusey CD. Pulmonary-renal syndrome: A life threatening but treatable condition. Postgrad Med J. 2013 May;89(1051):274–283.
- Saposnik G, Redelmeier D, Ruff CC, Tobler PN. Cognitive biases associated with medical decisions: A systematic review. BMC Med Inform Decis Mak. 2016 Nov 3;16(1):1–14.
- Guillevin L, Durand-Gasselin B, Cevallos R, et al. Microscopic polyangiitis: Clinical and laboratory findings in eighty-five patients. Arthritis Rheum. 1999 Mar;42(3):421–430.
- Jayne D. The diagnosis of vasculitis. Best Pract Res Clin Rheumatol. 2009 Jun;23(3):445–453.
- Jennette JC, Falk RJ, Bacon PA, et al. 2012 revised International Chapel Hill consensus conference nomenclature of vasculitides. Arthritis Rheum. 2013 Jan;65(1):1–11.
- Unizony S, Villarreal M, Miloslavsky EM, et al. Clinical outcomes of treatment of anti-neutrophil cytoplasmic antibody (ANCA)-associated vasculitis based on ANCA type. Ann Rheum Dis. 2016 Jun;75(6):1166–1169.
- Seo P, Stone JH. The antineutrophil cytoplasmic antibody-associated vasculitides. Am J Med; 2004 Jul 1;117(1):39–50.
- 1Berden AE, Ferrario F, Hagen EC, et al. Histopathologic classification of ANCA-associated glomerulonephritis. J Am Soc Nephrol. 2010 Oct;21(10):1628–1636.
- Hauer HA, Bajema IM, van Houwelingen HC, et al. Renal histology in ANCA-associated vasculitis: Differences between diagnostic and serologic subgroups. Kidney Int. 2002 Jan;61(1):80–89.
- Savige J, Pollock W, Trevisin M. What do antineutrophil cytoplasmic antibodies (ANCA) tell us? Best Pract Res Clin Rheumatol. 2005 Apr;19(2):263–276.
- Yates M, Watts RA, Bajema IM, et al. EULAR/ERA-EDTA recommendations for the management of ANCA-associated vasculitis. Ann Rheum Dis. 2016 Sep;75(9):1583–1594.
- Jones RB, Cohen Tervaert JW, Hauser T, et al. Rituximab versus cyclophosphamide in ANCA-associated renal vasculitis. N Engl J Med. 2010 Jul 15;363(3):211–220.
- Walsh M, Merkel PA, Peh C-A, et al. Plasma exchange and glucocorticoids in severe ANCA-associated vasculitis. N Engl J Med. 2020 Feb 13;382(7):622–631.