Discussion
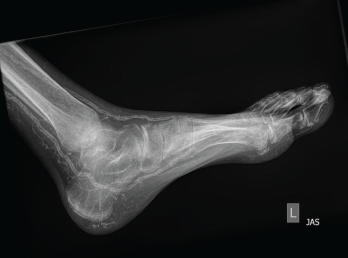
Figure 2: An X-ray of the left foot showed extensive vascular calcifications and no radiographic evidence of gout.
Crystalline arthropathies, such as gout, calcium pyrophosphate deposition (CPPD) disease and hydroxyapatite deposition, are not uncommon in patients with end-stage renal disease. The gold standard for crystalline diagnosis remains synovial fluid aspiration demonstrating the specific crystal, but in this case there was no joint effusion present for aspiration. The presence of monosodium urate (MSU) or CPPD crystals can be seen under polarized microscopy upon routine synovial fluid analysis, but other calcium crystals (e.g., hydroxyapatite and oxalate) are not typically reported in standard laboratory synovial fluid analysis and require additional stains (e.g., alizarin red) to distinguish crystalline etiology. Without confirmation of synovial fluid analysis, these crystal arthropathies can prove difficult to clinically distinguish from one another.
Gout is incredibly common in end-stage renal disease and is often easier to identify earlier in the course of the disease when it involves the first metatarsophalangeal joint. In renal failure, and as gout progresses, it can present as polyarticular and can be symmetric, involving the midfoot, ankle, knee and elbow.2
CPPD disease, often considered a gout mimic, has multiple presentations, from acute CPPD arthritis (similar to gout attacks) to osteoarthritis with CPPD to chronic inflammatory arthritis (i.e., a pseudo-rheumatoid arthritis presentation). It can coexist with gout, septic arthritis, osteoarthritis, rheumatoid arthritis and oxalate arthropathy.3,4
Oxalic acid arthropathy is an infrequent arthritic manifestation of hyperoxaluria that is associated with calcium oxalate deposition in tendons, cartilage, synovium and even blood vessels, as in our patient.5 Oxalate is primarily excreted through the kidney, and therefore hyperoxaluria can be the result of decreased excretion from renal failure. vitamin C supplementation can exacerbate it.6
Renal oxalosis occurs in a large majority of patients requiring long-term hemodialysis, and concentrations as high as 30 mcmol/L are not uncommon.3 The resulting inflammation can manifest as a symmetric polyarthritis that typically involves the PIP, metacarpophalangeal joints, knee, elbow and ankle joints.3 Deposition in blood vessels can further confound the picture because it can mimic vasculitis when severe.5
Important: The gold standard for oxalic acid arthropathy diagnosis is demonstration of positively birefringent crystals within synovial fluid or tissue that stain positive with alizarin red.
Imaging can help crystalline disease diagnosis when synovial fluid is not available for aspiration. The ACR/EULAR classification criteria for gout include use of imaging with ultrasound or dual-energy computed tomography (DECT) to identify the presence or absence of a double contour sign on ultrasound or urate deposition on DECT, and/or radiographic evidence of gout-related joint damage (e.g., gout-related erosion with a cortical break with a sclerotic margin and overhanging edge, excluding the gull wing appearance typically seen in erosive osteoarthritis).7
Ultrasound and CT also can demonstrate chondrocalcinosis, the deposition of CPPD in hyaline and fibrocartilage. The presence of chondrocalcinosis, however, can be an incidental finding and does not provide definitive proof that CPPD is causing the joint pain.7,8
No data exists on use of ultrasound or DECT to distinguish oxalate arthropathy. There is research interest on altering current DECT algorithms to try to differentiate calcium hydroxyapatite from CPPD. Radiographs of joints affected by these less common crystal arthropathies often have nonspecific radiographic findings such as vascular calcifications, which can also appear in renal osteodystrophy and chronic kidney disease and therefore do not confirm the diagnosis.9 This overlap of associated radiographic findings makes it difficult to determine if a finding is related to the primary pathology causing a patient’s symptoms, or if it is incidental and without clinical correlation.
In our case, the patient’s oxalate level was severely elevated—in a range more commonly seen in primary hereditary oxalosis and in which organ and joint involvement is almost universal.3,10 Given that there was no effusion to aspirate and that a tissue biopsy would not change management, these interventions were deferred.
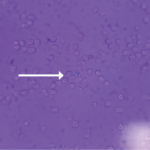
Without crystal-proven fluid/tissue analysis, it’s difficult to say for certain his symptoms were solely attributed to hyperoxaluria because CPPD, hydroxyapatite arthropathy and renal osteodystrophy can all contribute to calcium deposition and arthropathy. However, the pattern of DIP joint involvement is less common with hydroxyapatite arthropathy or renal dystrophy, and CPPD would not explain the vascular calcifications. The lack of MSU and the appearance of CPPD crystals on ultrasound also lowered our suspicion for gout and CPPD.
The most effective management for oxalate arthropathy is preventive, with the removal of oxalate through hemodialysis and limiting ascorbic acid intake. However, even with hemodialysis, removal is incomplete and often requires high flux membranes or daily hemodialysis.3 Systemic or intra-articular steroids remain the mainstay therapy for acute oxalate arthritis in patients with renal failure. In the absence of renal dysfunction, such as in hereditary or primary hyperoxaluria, non-steroidal anti-inflammatory drugs and colchicine can be used.
Data exist indicating the inflammatory pathways in oxalic acid arthropathic are interleukin 1β mediated, suggesting
IL-1 inhibition may be another option in the future.11