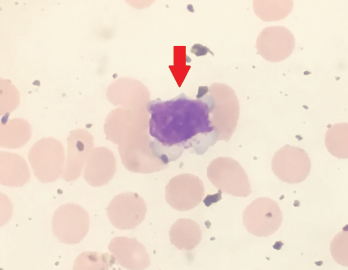
Figure 1. Peripheral blood smear showing a large granular lymphocyte in a 53-year-old Hispanic man.
Large granular lymphocytic (LGL) leukemia is a rare, chronic, lymphoproliferative disorder of cytotoxic T cell or natural killer cell lineage with an annual incidence of 0.72 cases per 1 million people in the U.S.1 The most common subtype of LGL leukemia, T-LGL leukemia, follows an indolent disease course and accounts for approximately 85% of cases. A second subtype, the chronic, lymphoproliferative disorder of natural killer cells (CLPD-NK), accounts for less than 10% of cases. The third subtype, known as aggressive natural killer-LGL leukemia, accounts for less than 5% of cases and carries a poor prognosis because it is refractory to chemotherapy.2,3
The diagnosis is made by first identifying an increased population of circulating large granular lymphocytic cells. An absolute lymphocyte count of >2,000 lymphocytes/mL may be helpful; however, it’s not mandatory because the diagnosis can be made if a clonal large granular lymphocytic population is identified in the clinical setting of autoimmune disease, such as rheumatoid arthritis or cytopenias (e.g., neutropenia, anemia, thrombocytopenia).2,4
Rheumatoid arthritis (RA) is one of the most common autoimmune disorders in developed countries, with prevalence estimates of approximately 0.5–1%.5 This chronic, systemic, inflammatory disorder is characterized by symmetric polyarticular synovial inflammation and joint destruction. RA has long been associated with LGL leukemia. Up to one-third of patients diagnosed with LGL leukemia will have a concomitant diagnosis of RA.2,4
RA is a known risk factor for the development of osteoporosis and vertebral fractures; however, little is known about the risk of osteoporosis and fractures in LGL leukemia. Here, we present a case of a patient with RA complicated by the sudden onset of LGL leukemia, as well as the accelerated development of multiple vertebral fractures.
Case Presentation
A 53-year-old Hispanic man with a 15-year history of high-titer, rheumatoid factor positive, anti-citrullinated protein antibody (ACPA) positive, nonerosive RA was seen in the outpatient rheumatology clinic for an RA flare. He complained of tenderness and swelling of both wrists, the metacarpophalangeal and proximal interphalangeal joints. He also reported a decreased ability to perform activities of daily living.
The patient had a long history of treatment-resistant RA, with treatment failures including hydroxychloroquine, sulfasalazine, adalimumab, etanercept, abatacept and tocilizumab. The decision was made to initiate certolizumab for management of his RA.
Five weeks after the initiation of certolizumab, he developed a new, severe neutropenia, with an absolute neutrophil count of 300 cells/μL. A peripheral smear showed atypical, large lymphocytes (see Figure 1), as well as smudge cells.
