Case study: You are asked to see a frail 78-year-old man with osteoarthritis and Parkinson’s disease who was recently discharged from the hospital after internal fixation of an intertrochanteric hip fracture sustained in a fall from standing height. The lowest T score on bone mineral density measurement is –1.7 at the total hip. Review of his laboratory results is notable for a serum calcium of 7.9 mg/dL, and an estimated glomerular filtration rate (GFR) of 40 mL/min. What would you do next?
Why Consider Bisphosphonates in the Hip-Fracture Population?
Hip fractures are one of the most dreaded results of osteoporosis, affecting one in six white women over the course of their lifetime and resulting in more deaths than breast cancer.1 For those individuals who survive, the consequences of a fracture include a high risk of mobility impairment, new functional dependence, and substantially elevated risk of hospitalization that persists for at least 12 months.2,3 Some of this morbidity is related to the occurrence of additional fractures after the initial event; prospective cohort studies have shown that one in five patients will suffer another fracture in the two years following their hip fracture, with nearly one-third of these being second hip fractures.4
Despite this high risk of additional osteoporotic fractures, fewer than 20% of older adults with hip fracture receive treatment for osteoporosis. This low secondary prevention rate holds true in nearly all settings and countries examined. While fragmentation of care and misaligned incentives have been implicated in the low treatment rate, surveys of providers caring for these frail older patients have also cited concerns that the high mortality rate after hip fracture may not allow sufficient time for the fracture-reduction benefit to accrue. Further, providers have been uncertain of the safety and efficacy of bisphosphonates in patients with multiple comorbidities and functional impairment, since most osteoporosis registration trials included a healthier, postmenopausal osteoporosis population.5 Finally, concerns have been raised about the impact of bisphosphonates on fracture healing in the hip-fracture population.
This article will summarize the evidence supporting the treatment of hip-fracture patients with bisphosphonates, including the impact of these agents on the occurrence of additional fractures, mortality, and safety endpoints. Practical and cost-effectiveness considerations will also be discussed.
Evidence Supporting Use of Bisphosphonates after Hip Fracture
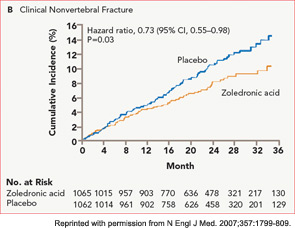
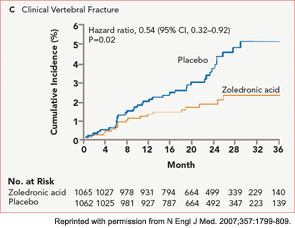
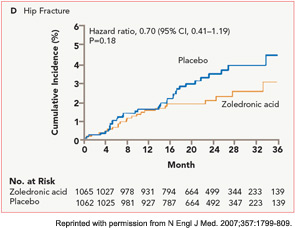
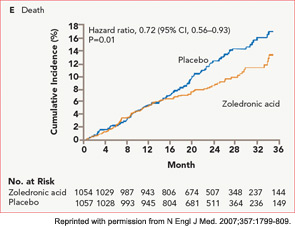
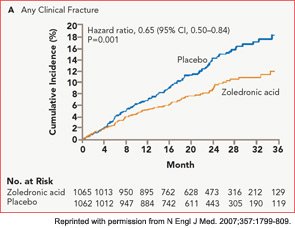
The HORIZON Recurrent Fracture Trial (HORIZON-RFT) was designed to determine the risks and benefits of bisphosphonate treatment in frail older adults with hip fracture. HORIZON-RFT was a randomized, controlled, double-blind study comparing zoledronic acid 5 mg IV yearly with a placebo. More than 2,100 patients were enrolled within 90 days of surgical repair of a low-trauma hip fracture, and were followed for up to three years for additional clinical fractures, bone mineral density (BMD), and safety endpoints including mortality (see Figure 1, above). Patients were enrolled from both community and institutional settings, but were ambulatory prior to the hip fracture. Other notable inclusion criteria were estimated GFR of greater than 30 mL/min, near-normal serum calcium levels, and inability or unwillingness to take an oral bisphosphonate. Because a large proportion of participants were found to be vitamin D insufficient, all participants received a loading dose of approximately 100,000 IU at least one week prior to the first dose of zoledronic acid, followed by daily oral calcium and vitamin D supplements. Patients were enrolled regardless of their baseline DXA, and 45% had T scores > –2.5.6