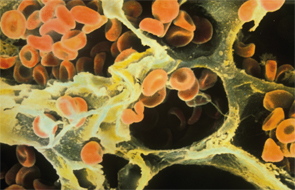
Red blood cells in the alveoli of a human lung on a scanning electron micrograph.
Image Credit: Ron Boardman/Life Science Image/FLPA/Science Source
Antiphospholipid syndrome (APS) is an autoimmune condition characterized by hypercoagulability often manifested as recurrent thrombosis or pregnancy complications, with persistently circulating antiphospholipid (aPL) antibodies or lupus anticoagulant. Catastrophic APS (CAPS), also known as Asherson syndrome, occurs in less than 1% of cases of APS and involves occlusive microangiopathy in at least three organ systems.1
Case Report
We report the case of a 43-year-old Guatemalan male with no significant past medical history who presented with a six-month history of progressive dyspnea, pleuritic chest pain and intermittent hemoptysis. On the day of presentation, he had sudden worsening of his cough and shortness of breath with frank hemoptysis. He had no bleeding or bruising at other sites. He also reported decreased appetite and increased fatigability, and had lost about 25 lbs. in the past year. He had no fever, chills or night sweats. There were no sick contacts, and he had not traveled recently. He had no history of prior surgical procedures and takes no medication except for the inhalers that were prescribed recently by another practitioner. There were no known drug allergies and no family history of cancer, bleeding disorders or other illnesses. He had a 10 pack-year history of cigarette smoking and quit about one month before presentation due to his progressive dyspnea. He used alcohol on the weekends, but did not use illicit drugs. He worked in a feather dye factory for 15 years, where he was exposed to sulfuric acid and ammonia.
On presentation, he was tachypneic, tachycardic, afebrile and normotensive, with no orthostatic change. In the emergency department, he was noted to have a cough productive of frothy bright red sputum. Oxygen saturation was 91% while breathing ambient air. His physical examination was significant for bilateral crackles and wheezing on lung examination and mild hepatomegaly. He had no lymphadenopathy. Initial investigations (see Table 1) revealed anemia, thrombocytopenia, mild leukocytosis, an activated partial thromboplastin time (aPTT) of 77.5 seconds, prothrombin time (PT) of 15.7 seconds and an international normalized ratio (INR) of 1.73. His electrocardiogram showed sinus tachycardia at a rate of 143 beats per minute without ST changes, and his chest radiograph showed bilateral predominantly perihilar alveolar opacities.
He was admitted and placed on contact isolation for suspicion of possible pulmonary tuberculosis. Hematology, infectious diseases and pulmonology subspecialties were consulted.
His condition improved following the infusion of intravenous crystalloids, broad-spectrum antibiotics and nebulization therapy. He was transfused with packed red blood cells and fresh frozen plasma, and was given Vitamin K. On Day 2 of his hospitalization, he underwent fiber-optic bronchoscopy that revealed mild diffuse erythema. Hemorrhagic bronchoalveolar lavage (BAL) fluid was sent for culture, cytology and tests for pneumocystis jirovecii and acid fast bacilli. Results were negative except for a gram stain showing many red blood cells, few white blood cells and no organisms. His respiratory status deteriorated, requiring invasive ventilatory support and critical care. Repeat chest radiograph showed worsening of the the alveolar opacities while his

(click for larger image)
Table 2: Patient Test Results for Systemic Causes of Hemoptysis
*Reference ranges are indicated in parentheses
**FEU = fibrinogen equivalent units
echocardiogram was suggestive of moderate mitral stenosis (valve area 1.5 square centimeters), severe tricuspid regurgitation and severe pulmonary hypertension with pulmonary artery systolic pressure of 74 millimeters of mercury. Computed tomography (CT) of the chest showed extensive alveolar consolidation. His blood and urine cultures were negative for growth.
Tests for possible disseminated intravascular coagulation (DIC), microscopic polyarteritis, Churg-Strauss syndrome, antiglomerular basement membrane antibody disease and granulomatosis with polyangiitis were negative (see Table 2). He had positive antinuclear antibody, anticardiolipin immunoglobulin M (IgM) and immunoglobulin G (IgG), elevated dilute Russell’s Viper Venom Time and aPTT mixing studies suggestive of a lupus anticoagulant (see Table 3). There was decreased factor VIII activity, which normalized with 1:20 dilution, also suggesting a lupus anticoagulant. During his hospital stay, it was discovered that he had scheduled foot surgery postponed because of a low platelet count and that he had tested positive for the lupus anticoagulant within the past year. Rheumatology was consulted, and he was started on high-dose corticosteroids.
His platelet count fell to a low of 24,000/cubic millimeters for which he received transfusions of platelets. He subsequently developed generalized tonic-clonic seizures and was started on anti-epileptics. CT of the head showed moderate to large acute infarction involving the left parietal-occipital cortex. He was started on cyclophosphamide and underwent many sessions of plasmapheresis, but continued to deteriorate. He expired 18 days after admission.

(click for larger image)
Table 3: Patient Test Results for Antiphospholipid Antibodies & Lupus Anticoagulant
*Reference ranges are indicated in parentheses
Discussion
For a male presenting with hemoptysis, thrombocytopenia and abnormal coagulation studies, a primarily prothrombotic disorder would not be immediately considered. Differentials for local and systemic disorders that can lead to hemoptysis would be entertained. Potential local causes include bronchopulmonary infections, such as bacterial pneumonia, pulmonary tuberculosis or bronchiectasis, as well as lung cancer, pulmonary edema and pulmonary embolism. Systemic causes, such as hemophilia, or an acquired coagulopathy, such as DIC, which would account for both deranged clotting function and thrombocytopenia, should be considered. In the case of diffuse alveolar hemorrhage, vasculitides, such as granulomatosis with polyangiitis, microscopic polyarteritis and Churg-Strauss syndrome, as well as anti-glomerular basement membrane antibody disease, should be considered.
Our patient’s clinical presentation and investigations are suggestive of primary APS manifested as probable CAPS. Although he had a positive ANA, he did not meet criteria for diagnosis of systemic lupus erythematosus (SLE). CAPS is a rare and devastating condition with protean manifestations. Diagnosis is based on the presence of persistent aPL antibody positivity, multi-organ involvement within a week and microthrombosis on histopathology (see Table 4, right).1,2 Presentation depends on the organ system involved and the degree of systemic inflammatory response. As such, diagnosis is particularly challenging in the initial phases when only one organ system is involved, a patient has no prior history of APS or presents with particularly unusual manifestations, such as bleeding.
The renal, pulmonary and central nervous systems are most commonly affected. Among the pulmonary manifestations, pulmonary embolism is most prevalent, but there have been reported cases of pulmonary hemorrhage.
Most cases of CAPS are associated with a precipitant, such as infection, surgery, subtherapeutic anticoagulation or SLE flares.1 More than one in three patients with CAPS will die. Thus, preventing CAPS and the aggressive treatment of cases are imperative.1
Anticoagulation with unfractionated or low molecular weight heparin for at least one week, followed by oral anticoagulants, is the mainstay of treatment.1 High-dose glucocorticosteroids are also used as a first-line intervention to contain the inflammatory response associated with CAPS. Treatment should continue for at least three days. In addition to steroids, plasmapheresis is used to curtail the inflammatory response by removing the antiphospholipid antibodies and inflammatory mediators. Likewise, intravenous immunoglobulin is also potentially useful, but it should be used with caution in patients who are concurrently bleeding because anticoagulants cannot be used to ameliorate its potentially prothrombotic effect. Other treatments include cyclophosphamide, which is found to be most useful in SLE-associated CAPS, and rituximab and eculizumab have also been used in limited cases.1,3
In the rare circumstance that a patient presents with bleeding, then immediate management should be targeted at ensuring a secure airway, maintaining oxygenation and ventilation and preventing circulatory collapse by adequate resuscitation using intravenous fluids and blood products as necessary. In addition to adequate resuscitation, attempts should be made to arrest bleeding by local control at the source and correcting coagulopathy. Of note, a prolonged aPTT in APS and CAPS is an in vitro manifestation of the lupus anticoagulant and indicates a tendency toward a procoagulant state. Therefore, utilizing the aPTT as the sole indication of a coagulopathy that needs correction is not useful in these patients. Instead, the complete picture inclusive of other coagulation studies should be taken into consideration. These include assessment of the PT/INR, thrombin time, factor assays, appropriate mixing studies, platelet count and platelet function tests, fibrinogen and fibrin degradation product levels and thromboelastography, where clinically appropriate.

More than one in three patients with CAPS will die. Thus, aggressive treatment is imperative.
Image Credit: Dragon Images/shutterstock.com
Thrombocytopenia is found in nearly half of patients with CAPS and would not account for bleeding until the count is less than 50,000/cubic millimeters. When present, concomitant DIC should be ruled out, because this condition would require transfusions of fresh frozen plasma and platelets. The benefit of transfusions of fresh frozen plasma in the bleeding patient with CAPS without any evidence of a factor deficiency or DIC has not been established, but should be considered in the case of large-volume blood transfusions because of the dilutional effect on clotting factors.
The precise mechanism by which APS causes pulmonary alveolar hemorrhage (PAH) has not been determined. In the largest case series of PAH in primary APS, lung biopsy specimen showed capillaritis and BAL fluid analysis revealed neutrophils and macrophages in keeping with an inflammatory cause of pulmonary hemorrhage. Anticoagulation, the first line in the treatment of CAPS, is contraindicated in these cases.
Conclusion
CAPS is rare, but it carries a high mortality rate. Pulmonary hemorrhage in particular is associated with a poor prognosis.4 Our case highlights the importance of increased awareness of the varied manifestations of this disease and the importance of aggressive treatment.

Dr. Tabanor
Joy-Ann Tabanor, MD, is the corresponding author, and a second-year resident physician in the Department of Medicine at Englewood Hospital and Medical Center in Englewood, N.J. She aspires to become a clinical rheumatologist.
Hyun Bae, MD, is a third-year resident physician in the Department of Medicine at Englewood Hospital and Medical Center in Englewood, N.J.
Girish Sonpal, MD, is a rheumatologist with decades of experience who practices in Whitestone, N.Y.
Karlene Williams, MD, is the associate program director for the Internal Medicine Residency Program at Englewood Hospital and Medical Center in Englewood, N.J.
References
- Cervera R, Espinosa G. Update on the catastrophic antiphospholipid syndrome and the ‘CAPS Registry.’ Semin Thromb Hemost. 2012 Jun;38(4):333–338. doi.
- Erkan D, Schur P. Diagnosis of the antiphospholipid syndrome. In: Pisetsky, D, ed. UpToDate. Waltham, Mass.: UpToDate; 2015. Accessed April 14, 2015.
- Shapira I, Andrade D, Allen SL, et al. Brief report: Induction of sustained remission in recurrent catastrophic antiphospholipid syndrome via inhibition of terminal complement with eculizumab. Arthritis Rheum. 2012 Aug;64(8):2719–2723.
- Cartin-Ceba R, Peikert T, Ashrani A, et al. Primary antiphospholipid syndrome-associated diffuse alveolar hemorrhage. Arthritis Care Res (Hoboken). 2014 Feb;66(2):301–310.