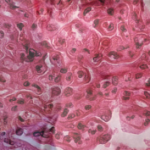
Hydroxychloroquine and naproxen controlled her symptoms for the next six months, and the anti-dsDNA and complement levels remained abnormal during this time. Microscopic hematuria along with 1 gram of urinary protein excretion was noted and confirmed on routine follow-up in the absence of symptoms. The serum creatinine and blood pressure were normal. Renal biopsy showed global, active diffuse proliferative nephritis (WHO Class IV), with an activity index of 13/24 and a chronicity index of 0/12; wire loops and crescent formation were seen. Pulse steroids followed by prednisone 1 mg/kg and MMF 3,000 mg/day were prescribed.
A year later, the renal disease had remitted, with preservation of renal function, normalization of serum complements and dsDNA. Prednisone was stopped and azathioprine substituted for MMF because the patient wanted to become pregnant.
Renal and extrarenal manifestations remained in remission for the next three years, during which she became pregnant and delivered a healthy daughter. Azathioprine was continued throughout the pregnancy and stopped at the patient’s request when she began breastfeeding. Surveillance serological studies during the pregnancy showed modest elevations of the dsDNA, intermittently low complements, without any clinical manifestations or other laboratory abnormalities.
Eighteen months following delivery, microscopic hematuria, along with an increase of the urinary protein excretion to 700 mg/24 hr, was noted on routine testing in the absence of symptoms, rise in blood pressure or worsening serum creatinine. Prednisone 20 mg/day and MMF 3,000 mg/day were restarted, with normalization of all urinary findings, preservation of GFR and continued modest elevations in repeated measurements of the anti-dsDNA and hypocomplementemia.
Limitations in Diagnosis
- Elevated anti-dsDNA often suggests a diagnosis of SLE, but it is an inadequate predictor of disease onset or flare. In the 1960s, it was shown that a rise in anti-dsDNA Ab levels along with a fall in complement levels predicted a flare of lupus nephritis.11 This correlation has held up fairly well, but not perfectly, and clearly something better is needed. This is illustrated in the case example, in which some correlation between anti-dsDNA and active nephritis was observed.
- No accurate peripheral blood cell and immune parameter predicts adequately intrarenal immunopathology.
- There are no reliable urinary biomarkers of active inflammatory intrarenal disease and renal fibrosis.
- There is a modest correlation between high anti-C1q antibodies and the development of LN (these antibodies were not measured in this patient).
Limitations in Our Understanding of Pathogenesis of LN
- What is it that determines which lupus patients develop renal disease?
- What is it that determines which lupus patients develop WHO Classes 2, 3, 4, 5, 6, and why then do some patients switch classes?
- Why do patients with LN tend to make complement fixing antibodies to dsDNA more so than patients without LN?
- Which of the genes that have been associated with lupus lead to LN and its variants (e.g., WHO Classes 2–6)?
- Are murine models of lupus applicable to human SLE? CTLA4-Ig acts synergistically to halt LN in murine models of lupus, but trials of abatacept and cyclophosphamide in humans showed no added benefit of abatacept.12 It’s not known whether features of trial design or intrinsic differences between human and animal disease accounted for this observation.
- What are the inflammatory pathways that lead to fibrosis, the pathologic determinant of ESRD, and can the transition from inflammation to fibrosis be interrupted?

Limitations in Treatment
- Current clinical trials are powered to detect primary outcome measures over short durations (6–12 months), which may not be clinically meaningful over the long term. (The index patient had an initial clinical response, but recurrence within four years of an apparent remission.) The NIH cyclophosphamide trial showed that it took about 10 years to clearly demonstrate that steroids plus cyclophosphamide was better than steroids alone for the prevention of renal failure.
- Racial and ethnic characteristics affect the responses to MMF; among black and Latino patients in the Aspreva Lupus Management (ALMS) study, 60% responded to MMF compared with 39% of patients treated with cyclophosphamide.13
- The ELN protocol was initially studied in Caucasians and, thus, not generalizable to patients of Hispanic, black or Asian descent, who are especially prone to LN. Insights regarding the comparable efficacy of this regimen in blacks and Hispanics with Caucasians was gleaned in the randomized trial of abatacept combined with the European cyclophosphamide regimen vs. placebo combined with the European regimen in patients with Class III/IV nephritis.14
- Murine models of disease have demonstrated a response to biologics (e.g., abatacept with CTX), which was not seen in humans, suggesting that more effective therapies might be better developed by a deeper understanding of human pathology.
- Which medication(s) are best to control blood pressure?
- What is the mechanism of action of ACE/ARB medications in reversing proteinuria? Answers to this question, as well as the preceding question, are largely extrapolated from studies of ACE inhibitors in diabetics and populations other than lupus.
- The appropriate duration of maintenance treatment with steroids, immunosuppressives and antihypertensives is unclear (index patient had renal flare within 18 months of stopping azathioprine).
- What treatment/medications will prevent renal fibrosis, prevent progression of fibrosis and reverse renal fibrosis?
The incremental progress in renal and patient survival amongst patients with LN over the past 60 years is related to effective treatment regimens informed by studies showing the remittive effects of corticosteroids alone followed by trials of immunosuppressive agents combined with steroids.