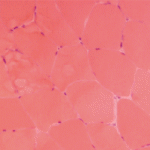
Michele Kaufman is a freelance medical writer based in New York City.
References
- Harper CR, Jacobson TA. The broad spectrum of statin myopathy: From myalgia to rhabdomyolysis. Curr Opin Lipidol. 2007; 18:401-408.
- Koumis T, Nathan JP, Rosenberg JM, Cicero LA. Strategies for the prevention and treatment of statin-induced myopathy: Is there a role for ubiquinone supplementation. Am J Health-Syst Pharm. 2004; 61:515-519.
- Pasternak RC, Smith C Jr, Bairney-Merz CN, Grundy SM, Cleeman JI, Lenfant C. ACC/AHA/NHLBI Clinical Advisory on the Use and Safety of Statins. Stroke. 2002;33:2337-2341.
- Antons KA, Williams CD, Baker SK, Phillips PS. Clinical perspectives of statin-induced rhabdomyolysis. Am J Med. 2006;119:400-409.
- Law M, Rudnicka AR. Statin Safety: A systematic review. Am J Cardiol. 2006;97(Suppl):52C-60C.
- Slim H, Thompson PD. Ezetimibe-related myopathy: A Systematic review. J Clin Lipidol. 2008;2:328-334.
- Christopher-Stine L. Stain myopathy: An update. Curr Opin Rhematol. 2006;16:647-653.
- Evans M, Rees A. Effects of HMG-CoA reductase inhibitors on skeletal muscle – Are all statins the same? Drug Safety. 2002;25:649-663.
- Klopstock T. Drug-induced myopathies. Curr Opin Neurol. 2008;21:590-595.
- McKenney JM, Davidson MH, Jacobson TA, Guyton JR. Final conclusions and recommendations of the National Lipid Association Statin Safety Assessment Task Force. Am J Cardiol. 2006;97(suppl):89C-94C.
- Link E for the SEARCH Collaborative Group. SCLO1B1 variants and statin-induced myopathy – A genomewide study. N Engl J Med. 2008;359:789-799.
- UCB files for R.: www.ucb-group.com/news/3019.asp. Last accessed October 20, 2008.
- FDA approves Cimzia to treat Crohn’s disease. www.fda.gov/bbs/topics/NEWS/2008/NEW01821.html. Last accessed October 20, 2008.
- EULAR RAPID results: www.medicalnewstoday.com/articles/111632.php Last accessed October 20, 2008.
- Additional RA indications/RA in Europe: http://hugin.info/133973/R/1239755/265632.pdf Last accessed October 20, 2008.
- BLA submitted for Golimumab. www.drugs.com/nda/golimumab_080627.html. Last accessed October 20, 2008.
- Drug Watch. Adult and juvenile rheumatoid arthritis drugs in development. Formulary. 2008;43:260.
- FDA issues complete response letter to Roche for ACTEMRA (tocilizumab) biologics license application. www.rocheusa.com/newsroom/current/2008/pr2008091801.html. Last accessed November 4, 2008.
- Important drug warning new safety information. www.fda.gov/medwatch/safety/2008/rituxan_DHCP_Final%209411700.pdf. Last accessed October 20, 2008.
- Prescribing information. www.fda.gov/medwatch/safety/2008/Rituxan_PI.pdf. Last accessed October 20, 2008.
- FDA: manufacturers of TNF-blocker drugs must highlight risk of fungal infections. www.fda.gov/bbs/topics/NEWS/2008/NEW01879.html. Last accessed October 20, 2008.
- Information for healthcare professionals Cimzia (certolizumab pegol), Enbrel (etanercept), Humira (adalimumab), and Remicade (infliximab) www.fda.gov/cder/drug/InfoSheets/HCP/TNF_blockersHCP.htm. Last accessed October 20, 2008.
- FDA approves updated labeling for psoriasis drug Raptiva. www.fda.gov/bbs/topics/NEWS/2008/NEW01905.html. Last accessed October 21, 2008.
- Prescribing information. www.gene.com/gene/products/information/pdf/raptiva-prescribing.pdf. Last accessed October 21, 2008.
- Important safety information. www.gene.com/gene/products/information/pdf/raptiva_dhcp.pdf. Last Accessed accessed October 21, 2008.