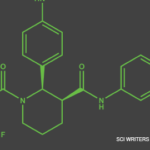
On Oct. 8, ChemoCentryx Inc. announced that the U.S. Food & Drug Administration (FDA) has approved avacopan (TAVNEOS), an orally administered selective complement 5a receptor inhibitor, as an adjunctive treatment of adult patients with severe active anti-neutrophil cytoplasmic autoantibody-associated vasculitis (ANCA-associated vasculitis), specifically granulomatosis with polyangiitis (GPA) and microscopic polyangiitis (MPA) (the two main forms of ANCA vasculitis), in combination with standard therapy. ANCA-associated vasculitis is a systemic autoimmune disease in which over-activation of the complement system further activates neutrophils, leading to inflammation and eventual destruction of small blood vessels. This results in organ damage and failure, with the kidney as the major target, and is often fatal if not treated.
“We look forward to making TAVNEOS available to clinicians and patients in the next few weeks,” said Thomas J. Schall, PhD, president and chief executive officer of ChemoCentryx.
“I am excited that our work has helped lead to the first-in-a-decade approval of a medicine for ANCA-associated vasculitis. This is an important step forward in the treatment of this disease,” said the trial’s co-primary academic investigator Peter A. Merkel, MD, MPH, the chief of rheumatology at the Perelman School of Medicine at the University of Pennsylvania, director of the international Vasculitis Clinical Research Consortium, and consultant to ChemoCentryx. “Patients will now have access to a new class of medication that provides beneficial effects for the treatment of ANCA-associated vasculitis.”
“The vasculitis community is elated that TAVNEOS is now approved, bringing a much-needed new treatment option to patients living with this devastating disease,” said Joyce Kullman, executive director, Vasculitis Foundation. “There is a significant unmet need in the treatment of ANCA-associated vasculitis, with current therapies often leading to serious, even fatal, side effects and a diminished quality of life. We believe new therapies like TAVNEOS may offer a brighter future for these patients.”
Avacopan is the first FDA approved orally administered inhibitor of the complement C5a receptor. The approval in ANCA-associated vasculitis was supported by the results of the pivotal phase 3 ADVOCATE trial, which were highlighted in the February 2021 edition of The New England Journal of Medicine (NEJM). The ADVOCATE trial of avacopan was a global, randomized, double-blind, active-controlled, double-dummy phase 3 trial of 330 patients with ANCA-associated vasculitis in 20 countries. Eligible study subjects were randomized to receive either rituximab or cyclophosphamide (followed by azathioprine/mycophenolate) and either avacopan or study-supplied oral prednisone. Subjects in both treatment groups could also receive non-protocol glucocorticoids if needed. The study met its primary endpoints of disease remission at 26 weeks and sustained remission at 52 weeks, as assessed by the Birmingham Vasculitis Activity Score, or BVAS. The study demonstrated superiority to a prednisone-based standard of care with respect to sustained remission at 52 weeks. The most common adverse reactions (≥5% of patients and higher in the avacopan group vs. prednisone group) were: nausea, headache, hypertension, diarrhea, vomiting, rash, fatigue, upper abdominal pain, dizziness, blood creatinine increase, and paresthesia.