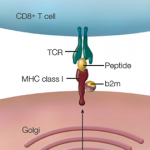
Yet these are all relatively straightforward adaptations of this technology. What happens when we start using CRISPR as a tool for human genome editing?1 For example, to treat human immunodeficiency virus (HIV) infection, physicians might edit a patient’s immune cells to delete the CCR5 gene, conferring the resistance to HIV carried by the 1% of the U.S. population lacking functional copies of this gene. For patients becoming progressively blind due to the genetically dominant forms of retinitis pigmentosa, the mutant allele in retinal cells might be inactivated. Patients at high risk for cardiovascular events because they are homozygous for familial hypercholesterolemia may one day undergo liver cell editing that could restore a functional copy of the gene encoding low-density lipoprotein receptors. Similarly, editing of blood stem cells might one day cure sickle cell anemia and hemophilia. Could harmful genes, such as BRCA, that confer a heightened risk for cancer be deleted from a child carrying this repertoire? Aldous Huxley must be rolling over in his grave.
Driving Genes
For all the benefits that CRISPR promises, there are some significant consequences to the use of this technology that society must consider carefully. Usually, a genetic change in one organism takes a long time to spread through a population. In 2003, an evolutionary biologist, Austin Burt, PhD, professor of evolutionary genetics at the Imperial College in London, England, outlined an ingenious way to flout the laws of both inheritance and evolution.7 For more than a century, geneticists have known that in organisms that pair up to reproduce, most genes have a 50–50 chance of being inherited, as Moravian scientist and Augustinian friar Gregor Mendel famously demonstrated using pea plants. Similarly, evolutionary biologists have known since Charles Darwin that natural selection eliminates inherited traits that reduce an organism’s fitness, such as a mosquito’s susceptibility to insecticide.
Dr. Burt described how gene drive can outsmart both Mendel and Darwin. In nature, he noted, some genes copy themselves into multiple places in a genome. Like a dishonest candidate stuffing the ballot box, these extra copies increase a gene’s odds of “winning,” that is, being inherited. In addition, other genes can destroy related ones, also ensuring their transmission into the next generation.
Although this natural gene drive is unpredictable and scattershot, Dr. Burt showed that certain tricks of molecular biology could achieve the same results—causing a gene to be inherited by many more organisms through many more generations than standard genetics and natural selection allow. His theory, published in 2003, lay dormant because there was no tool available that could put this theory to practice. But now, instead of genetically engineering single organisms, gene drive can sweep an edited gene exponentially faster through a population by allowing a mutation made by CRISPR on one chromosome to copy itself to its partner in every generation, so that nearly all offspring will inherit the change. The genetics of vast populations are immediately altered. For example, a mutation engineered into a mosquito strain that carries the malaria parasite could spread through a large population within a season. If that mutation reduced the number of offspring a mosquito produced, then the population could be wiped out, along with any malaria parasites being carried. In fact, a recent report described a working prototype for this experiment.8