(Reuters Health)—The live herpes zoster vaccine does not provide reliable long-term protection in rheumatoid arthritis (RA) patients taking tofacitinib, a recent study suggests.
Current ACR guidelines conditionally recommend that patients with RA who are 50 years and older be vaccinated against herpes zoster prior to starting therapy with the Janus kinase (JAK) inhibitor tofacitinib or with biologic disease modifying antirheumatic drugs (bDMARDs). Herpes zoster incidence is approximately twofold higher with tofacitinib as it is with bDMARDs, the study team notes.
For the current analysis, researchers looked at data on 100 RA patients who took tofacitinib after receiving the live herpes zoster vaccine, before the recombinant zoster vaccine became available in 2017. This included 46 people on an average dose of 5 mg and 54 people on an average dose of 10 mg two times per day.
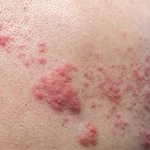
Five patients developed herpes zoster, at 218, 280, 748,741, and 544 days post-vaccination, respectively. All cases were mild to moderate in severity and resolved with antiviral treatment.
Three of the five patients who developed herpes zoster during the study had undetectable varicella zoster virus (VZV) cell-mediated immunity after vaccination, both at baseline and at week 6.
The other two patients who developed herpes zoster responded adequately to vaccination by both IgG and ELISPOT measures, but had lower than average VZV IgG levels at baseline and Week 6, the study team writes in Annals of the Rheumatic Diseases.1
“The study suggests the live vaccine is likely less protective in rheumatology patients who subsequently start JAK inhibitors as compared to the general population,” says lead study author Kevin Winthrop, MD, MPD, of Oregon Health & Science University School of Medicine, Portland.
“RA patients build less robust cell-mediated immunity after vaccination with this live vaccine as compared to the general population,” Dr. Winthrop says by email. “It is likely that less robust cellular immunity translates to less robust short-term and long-term protection.”
Although it’s possible that LZV booster vaccinations may improve vaccine efficacy, to date there is a lack of data on the use and timing of booster vaccinations, and no recommendations on the use of LZV booster vaccinations currently exist, the study team writes.
This highlights the importance of evaluating the newly approved subunit non-live vaccine (Shingrix) in patients with RA receiving tofacitinib, the authors write. The study was sponsored by Pfizer, maker of tofacitinib, and several authors on the study have received a variety of funding from Pfizer and several other drug companies.