 ACR BEYOND LIVE—Among patients with systemic lupus erythematosus (SLE), lupus nephritis remains one of the leading causes of mortality, and patients with both SLE and end-stage renal disease demonstrate standardized mortality ratios higher than 60 times that of patients with SLE who have normal kidney function.1 Although the ACR Guidelines for Screening, Treatment, and Management of Lupus Nephritis, published in 2012, appear to be straightforward in terms of choices for induction and maintenance therapies, the complex biology and heterogeneous phenotypes of lupus nephritis make the story more complicated.2
ACR BEYOND LIVE—Among patients with systemic lupus erythematosus (SLE), lupus nephritis remains one of the leading causes of mortality, and patients with both SLE and end-stage renal disease demonstrate standardized mortality ratios higher than 60 times that of patients with SLE who have normal kidney function.1 Although the ACR Guidelines for Screening, Treatment, and Management of Lupus Nephritis, published in 2012, appear to be straightforward in terms of choices for induction and maintenance therapies, the complex biology and heterogeneous phenotypes of lupus nephritis make the story more complicated.2
At the 2020 ACR State-of-the-Art Clinical Symposium, Joan Merrill, MD, director of clinical projects in the Arthritis & Clinical Immunology Research Program at the Oklahoma Medical Research Foundation, Oklahoma City, discussed updates in the understanding and treatment of lupus nephritis.
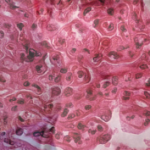
Dr. Merrill began her talk by noting several baseline characteristics of patients with lupus nephritis that may portend a poor prognosis, such as non-European ancestry, young age, elevated serum creatinine or chronicity index, class IV lesions on biopsy, and anti-Ro antibodies. In particular, she stated that it’s important to pay attention to tubulointerstitial involvement on renal biopsy, because it may be a significant indicator of high risk for progression to renal failure.3
Many rheumatologists are aware of such options as cyclophosphamide or mycophenolate mofetil as induction treatment for lupus nephritis, but the most exciting areas of research in lupus nephritis include the evolving comprehension of how factors specific to each patient and their expression of disease may guide treatment. For example, the 2012 ACR guidelines point out that mycophenolate mofetil is preferred over cyclophosphamide as induction therapy for class III and IV lupus nephritis in African American and Hispanic patients.2
A randomized trial of multi-target induction therapy with tacrolimus plus mycophenolate mofetil vs. cyclophosphamide alone in patients with lupus nephritis across 26 renal centers in China showed multi-target treatment was superior in this population, and this has had important implications for use of calcineurin inhibitors in Asian patients with lupus nephritis.4
Partnership for Drug Targets
On a molecular level, researchers since 2003 have been investigating the prominent expression of interferon (IFN) regulated genes—an IFN signature—in the blood and tissues of patients with SLE, prompting such questions as: What is the cause of the IFN signature? Which cells produce IFN and does this change over different phases of the disease? Should treatment focus on blocking the IFN system in SLE and, if so, which is the most suitable target?5