Javier Regueiro / shutterstock.com
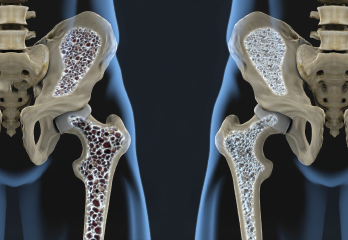
CHICAGO—The osteoporosis session at the 2018 ACR/ARHP Annual Meeting opened with a discussion by Kenneth Saag, MD, MSc, professor of medicine at the University of Alabama, Birmingham, in which he highlighted adverse events associated with osteoporosis medications.
Dr. Saag began his presentation by emphasizing that, above all, the audience should keep in mind that the biggest crisis in osteoporosis is the decline in treatment for both the primary and secondary prevention of osteoporosis. He reminded the audience to place his talk in this context and recognize that all drugs have potential adverse effects.
Bisphosphonates Risk
Healthcare providers prescribe many different drugs to treat osteoporosis, and although Dr. Saag touched on all of them, he spent most of his time focusing on bisphosphonates, answering the question, “Is it bad for the bone and other tissues?” He emphasized the benefits of bisphosphonate treatment are clear and described them as the “gift that never stops giving,” noting that if a patient stops taking alendronate after a 10-year period, the drug continues to persist in the bone. That persistence means the patient continues to receive the bioequivalence of approximately 25% of the daily dose.
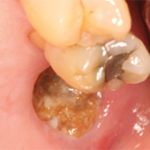
Unfortunately, some patients who take bisphosphonates may also experience osteonecrosis of the jaw (ONJ), which presents as an area of exposed and/or necrotic bone in the maxillofacial region. This very rare side effect can prove quite serious, especially if the patient experiences an even less common pathologic jaw fracture. Dr. Saag explained it may be possible to prevent ONJ prior to anti-resorptive therapy by treating oral infection with antibiotics. He also noted the importance of ensuring patients realize that if they plan to have dental work done, that work should be healed prior to when they start anti-resorptive therapy.
Bisphosphonates can also cause atypical femur fractures (AFF), which form along the femoral diaphysis from lesser trochanter to supracondylar. Such fractures are sometimes described as the dreaded black line. The risk of an AFF among bisphosphonate users is estimated to be eight in 10,000. Additional possible side effects, such as esophageal and gastric cancer, have some biologic rationale, Dr. Saag explained, and are less well understood and even less likely to be causally related to bisphosphonate use.
Give Them a Break
Given the clear but rare adverse events associated with bisphosphonate use, a physician must determine for each patient if the known benefits outweigh the potential risks. Dr. Saag suggested that to manage the risk-benefit ratio, rheumatologists should query patients for signs of AFF via patient history and physical examination. This is best performed by focusing on new hip or thigh pain. Patients with thigh pain associated with a stress reaction or incomplete AFF may require prophylactic surgery and rodding. They may also need to take a break from bisphosphonates. Dr. Saag explained noted that, “We may want to rest and reenergize and consider switching to something different after that period of time.” Such a break may be as short as one year.
The American Society for Bone and Mineral Research (ASBMR) has put forward an algorithmic approach to taking such a break from bisphosphonates. Analyses of U.S. Medicare data, as well as other large datasets, indicate the risk of AFFs decline after a drug holiday of one year. This is encouraging, especially because AFF and ONJ are the adverse events most certainly associated with bisphosphonates.
Dr. Saag concluded his presentation by stating the use of osteoporosis drugs requires careful and recurrent assessment of fracture benefits and adverse event risks. As part of that assessment, he encouraged the audience to seriously consider the possibility that patients on bisphosphonates should receive a drug holiday or switch to another treatment.
Healthcare providers prescribe many different drugs to treat osteoporosis, including bisphosphonates.
Kidney Disease & Fracture Risk
Susan Ott, MD, professor of medicine and metabolism at the University of Wisconsin School of Medicine in Madison, discussed bone health in patients with chronic kidney disease (CKD) who have high fracture risk that presents throughout all stages of disease.
These patients have increased bone turnover due to multiple factors associated with CKD, Dr. Ott explained. Patients with renal failure have difficulty excreting phosphate, a situation that affects bone metabolism because calcium and phosphate are in a complex feedback loop with parathyroid hormone (PTH) and vitamin D.
Patients with CKD also have low levels of bone morphogenic protein 7 (BMP-7), which is necessary for differentiation of pre-osteoblasts to mature osteoblasts. This loss of BMP-7 causes the histologic finding known as fibrosis. In addition, patients may be malnourished, which can contribute to decreased bone formation.
Patients with CKD may also experience the accumulation of iron, aluminum and strontium in the bone, which can contribute to osteomalacia and oxalate in the bone, which can cause crystal deposition. Moreover, dialysis does not clear beta-2 microglobulin, so patients on long-term dialysis can form amyloid that can subsequently create cysts and cause carpal tunnel syndrome. The cysts frequently form at the end of a long bone, and, when the diagnosis is missed by a nephrologist, the patient is frequently referred to a rheumatologist.
Dr. Ott explained that for patients with slightly low PTH, the best option may be treatment with raloxifene or possibly anti-sclerostin, when available. She also noted that most nephrologists treat patients with mild to moderate kidney disease with denosumab. Unfortunately, the safety of bisphosphonates in patients with decreased kidney function is unknown, and the effectiveness of teriperatide in patients with high PTH due to kidney disease is unknown.
“We have a really complex set of symptoms,” Dr. Ott concluded. “It will be hard to find treatments when there are so many things going wrong. You just take them one at a time, and do what you can.”
Lara C. Pullen, PhD, is a medical writer based in the Chicago area.