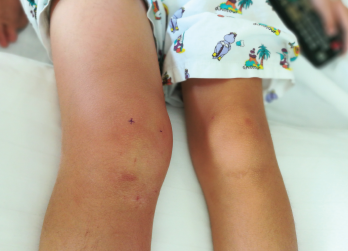
The right knee, swollen in comparison to the left, could be due to acute joint inflammation or infection.
DonyaHHI / shutterstock.com
ATLANTA—Managing pediatric patients with rheumatic disease involves special considerations, such as developmental concerns and physiological traits that may affect dosing of medications, according to two experts. During a session at the 2019 ACR/ARP Annual Meeting, Courtney Kremer, ARNP, a pediatric nurse practitioner at the University of Iowa Stead Family Children’s Hospital, Iowa City, and Jessica Lynton, PharmD, a clinical pharmacy specialist at the same center, discussed important aspects to remember when treating young patients. They also reviewed cases and their treatment plans.
“It’s a common misconception that pediatric patients will outgrow their rheumatologic disease,” Ms. Kremer said. “Approximately 50% of young adults with JIA [juvenile idiopathic arthritis] will have ongoing, active disease, and it’s not uncommon [for some patients to] have lifelong disease.”
Treatment Considerations
Treatment should be aggressive. When left unchecked, active inflammation can lead to growth disturbances, which have the potential to stunt a patient’s final height and may result in osteopenia and limb-length discrepancies, Ms. Kremer said. Aggressive treatment results in better overall outcomes.
When choosing a medication and its dosage, clinicians must consider variations in how it’s absorbed, Dr. Lynton said. The rate and extent of a patient’s organ function and development will affect the drug’s distribution, as well as how it’s metabolized and eliminated.
No simple rule exists for making alterations to adult dosing guidelines for pediatric patients. “The theme that’s important to remember is that the doses can’t just be halved or … decreased,” she said. “Doses are going to vary. And sometimes, those dose requirements can be greater than what we would expect in the adult population.”
Example: 78% of the body weight of a full-term infant is water, but it’s only 60% in adults. Thus, some medications will have a greater volume of distribution in infants and children. Body fat is also much lower in neonates than in adults. Therefore, drugs that are highly lipid soluble will be distributed less widely in infants than in adults.
Case Studies
Case 1: Ms. Kremer presented the case of 6-year-old girl who had a swollen knee for two months that was causing her to limp each morning. She was unable to straighten her right leg, and the knee was warm on physical exam. The girl had a history of camping and tick exposure, but no fevers or illness before the symptoms started, and no family history of autoimmune disease. She had a high erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) level, and was anti-nuclear antibody (ANA) positive. X-rays showed effusion, but no lesions or erosions.
For this patient, Ms. Kremer said trauma, septic arthritis, a bone tumor, inflammatory diseases, mechanical problems, infectious disease and hemarthrosis were all considered. Labs and imaging ruled out trauma, infection, tumor and other causes. Clinicians diagnosed the girl with oligoarticular JIA.
This disease is the most common subtype of JIA, making up 50% of cases, she said. Patients are typically female and present with a small number of swollen joints that are mostly in the lower extremities. Most patients are ANA positive and at high risk for eye inflammation.
The goal with treatment, clearly, is disease remission, Ms. Kremer noted. “This may require rapid medication escalation to gain adequate control. We want to prevent joint damage and maintain as much normal function as we can. And of course, we want to avoid medication toxicity,” she said.
Treatment options include triamcinolone acetonide injections, disease-modifying anti-rheumatic drugs (DMARDs), methotrexate, leflunomide, sulfasalazine and hydroxychloroquine.
For this patient, clinicians designed a treatment plan that included an intra-articular triamcinolone injection with 10 mg/kg per day of naproxen until the swelling and pain resolved. The dose of triamcinolone is 1–2 mg per kilogram per joint, depending on the size of the joint and the child, she said. The patient was prescribed physical therapy and asked to follow up in three months. If the knee swelling came back, clinicians planned to add a DMARD, possibly with another injection.
Additionally, because the girl was at a high risk for eye inflammation, she needed to undergo ophthalmology screening every three to four months.
Case 2: Another case involved a 15-year-old softball player who had right wrist and elbow pain that began during the previous softball season. She was experiencing increasing stiffness and trouble holding a toothbrush in the morning. She had no fevers or night sweats, no gastrointestinal symptoms and no pulmonary symptoms.
On exam, she had decreased wrist extension on both sides, decreased elbow extension, the inability to straighten her elbow and swelling in all of the small joints in her hands. She was anemic, positive for rheumatoid factor and ANA antibodies, and had elevated ESR and CRP levels.
In this case, the differential diagnosis included juvenile arthritis, lupus, dermatomyositis, vasculitis, scleroderma, inflammatory bowel disease, infections, immunologic or metabolic causes, malignancies, mechanical causes and pain syndromes.
A repeat test was also positive for rheumatoid factor, but the lab results did not meet the criteria for lupus. X-rays showed osteopenia of the MCP, PIP and DIP joints, with carpal crowding of the wrist and osteopenia of the distal, radial and ulnar joints.
She was diagnosed with rheumatoid factor-positive, polyarticular JIA.
“These [JIA] patients can be rheumatoid factor positive or negative—although it’s rare to see a positive rheumatoid factor in JIA,” Ms. Kremer said. “It accounts for less than 5% of patients. When positive, it’s considered the equivalent to the adult form of rheumatoid arthritis.”
In this form of JIA, joints are often involved symmetrically, with both large and small joints affected. Fewer than 50% of these patients go into remission without medications.
Along with the medications considered for the previous case, biologics are an option. If a patient doesn’t respond to one anti-tumor necrosis factor (TNF) drug, such as adalimumab, etanercept or infliximab, they may respond to another anti-TNF, she said.
Other treatment options include the CTLA-4 inhibitor, abatacept, and the IL-6-inhibitor, tocilizumab. All of these treatments require monitoring with complete blood count (CBC) and liver function tests. With tocilizumab, periodic lipid panels are also needed, Dr. Lynton noted.
Clinicians treated the patient in the second case with 0.5 mg/kg subcutaneous methotrexate weekly, as well as oral folic acid daily and 8 mg of ondansetron every eight hours for nausea, or as needed. At the same time, 0.8 mg/kg etanercept was given every week. The patient was on a non-steroidal anti-inflammatory drug (NSAID), with the plan of stopping it upon remission. Baseline testing and subsequent monitoring were needed for tuberculosis, hepatitis B, hepatitis C, serum creatinine and CBC with differential and liver function tests.
Biologics
Despite the increased care necessary to monitor such patients, the use of biologics has been a huge step forward in the field. Dr. Lynton showed a picture of a mother who didn’t have access to biologics at the start of her disease and had hands with permanent, disfiguring damage. She was seated next to her son with JIA, who had been treated early with biologics and had hands that appeared normal.
“Biologics have revolutionized the outcomes in polyarticular JIA,” said Dr. Lynton.
Thomas R. Collins is a freelance writer living in South Florida.

