Denis Cristo / shutterstock.com
AMSTERDAM—Molecular signatures in synovial tissue that can be gathered through biopsies are a largely untapped resource that could help guide treatment for rheumatic conditions, an expert said at the EULAR: Annual European Congress of Rheumatology.
“I’m convinced that looking into synovial tissue provides us with tools to not only understand mechanisms of disease in rheumatoid arthritis or psoriatic arthritis, [but] it might also give us a tool to make a clinical decision,” said Bernard Lauwerys, MD, PhD, professor of rheumatology at Université Catholique de Louvain in Brussels.
In a session on emerging biomarkers in psoriatic arthritis (PsA), Dr. Lauwerys said not much data has been amassed through molecular examination of synovial tissue in the disease. But insights into rheumatoid arthritis (RA) offer encouragement for what could emerge.
For example, examinations of cell populations before and after treatment with adalimumab in RA patients have found that several subtypes decrease, particularly CD68-positive cells. This offers up a “very good marker” for treatment effect, he said.1
Molecular profiling for different treatments shows some—but not all—treatments have roughly the same effect in RA patients, he said.
Rituximab, methotrexate and tocilizumab tend to have an overlapping effect, but the effect doesn’t overlap with adalimumab’s.2
“Those treatments do quite the same from a molecular point of view in synovial tissue, and this is a decrease in T cell activation,” Dr. Lauwerys said. “By contrast, there is really no common effect between adalimumab and any of those drugs. Whether a transcript is downregulated or upregulated by adalimumab doesn’t predict what will be the effect of tocilizumab.”
Such information can inform clinical decision making, he said.
The Studies
In a study of PsA and other spondyloarthritides, examination of tissue from 60 synovial biopsies found a common denominator in overexpression of a TGF-beta myogenic signature.3
In his group’s own unpublished evaluation of synovial tissue, “quite a bit of heterogeneity in molecular patterns” was found among patients with spondyloarthritides. But more work is needed to harness the power of this information, Dr. Lauwerys said. He hopes for the development of what he called a “molecular taxonomy,” groupings based on what these patterns are saying. The EULAR Synovitis Study Group is at work in this area.
“This will be interesting if we can link this to medical decisions,” he said.
Serum Biomarkers Needed
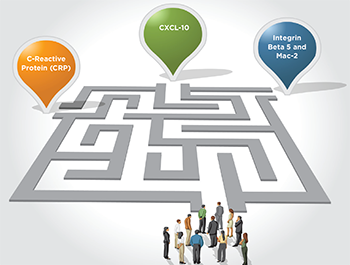
Christopher Ritchlin, MD, MPH, said the need for serum biomarkers in PsA is clear, noting the efficacy of one of the primary tools used now, C-reactive protein (CRP), is severely limited.
“We need biomarkers that are more informative than CRP because we know that only 40–50% of patients with PsA have elevated CRP and many patients with very active disease have no elevation in this marker,” he said.
One biomarker for risk of psoriasis progressing to PsA is the chemokine CXCL-10. It has been found to be significantly elevated in patients who convert from psoriasis to PsA. But research has also shown that, once a diagnosis of PsA is made, CXCL-10 levels actually decline, even among those who haven’t received treatment yet. The mechanisms that underlie this decline are not well understood.4
“This is a chemokine we need to better understand and may be prove valuable in the identification of psoriasis patients at increased risk for arthritis,” he said.
Other markers being evaluated seem to have the potential to improve diagnostic accuracy of PsA. When added to CRP, two molecules—integrin beta 5 and Mac-2 binding protein—improve the ability to make a PsA diagnosis.5
Noncoding microRNAs are another area in PsA likely to increase in importance, Dr. Ritchlin said.6 This is particularly relevant, because it is now known that much of the genetic risk for immune inflammatory disorders is in the noncoding regions at the sites of enhancer and promoter activity.
“Thinking about the noncoding region of the genome is very important as well for serum biomarkers and this is an area that I think is going to receive a lot more attention,” he said.
All of these biomarker candidates need to further explored and validated in a rigorous manner before they can be used, he said.
“To develop these kinds of biomarkers requires a lot of validation and confirmation in different disease cohorts,” he said. “We don’t have that level of validation at this time for the markers discussed today, but we do have several biomarkers that can be formally evaluated in clinical trials and observational cohorts.”
Thomas R. Collins is a freelance writer living in South Florida.
References
- Badot V, Galant C, Nzeusseu Toukap A, et al. Gene expression profiling in the synovium identifies a predictive signature of absence of response to adalimumab therapy in rheumatoid arthritis. Arthritis Res Ther. 2009;11(2):R57.
- Ducreux J, Durez P, Galant C, et al. Global molecular effects of tocilizumab therapy in rheumatoid arthritis synovium. Arthritis Rheumatol. 2014 Jan;66(1):15–23.
- Yeremenko N, Noordenbos T, Cantaert T, et al. Disease-specific and inflammation-independent stromal alterations in spondylarthritis synovitis. Arthritis Rheum. 2013 Jan;65(1):174–185.
- Abji F, Pollock RA, Liang K, et al. Brief Report: CXCL10 is a possible biomarker for the development of psoriatic arthritis among patients with psoriasis. Arthritis Rheumatol. 2016 Dec;68(12):2911–2916.
- Cretu D, Gao L, Liang K, et al. Differentiating psoriatic arthritis from psoriasis without psoriatic arthritis using novel serum biomarkers. Arthritis Care Res (Hoboken). 2018 Mar;70(3):454–461.
- Ciancio G, Ferracin M, Saccenti E, et al. Characterisation of peripheral blood mononuclear cell microRNA in early onset psoriatic arthritis. Clin Exp Rheumatol. 2017 Jan–Feb;35(1):113–121.