CNRI / Science Source
Renal arteriosclerosis is common in lupus patients with nephritis and occurs two decades earlier than it does in people without lupus nephritis (LN), report investigators in a study that examined the prevalence of renal arteriosclerosis in LN patients compared with healthy controls.1
The finding suggests that renal arteriosclerosis could be used as a biomarker for early prediction of cardiovascular disease (CVD) risk in LN patients and subsequently lead to timely CVD prevention.
Improving prevention of CVD in patients with LN is needed given the high risk of CVD in these patients. Compared with their healthy controls, patients with LN have a ninefold higher risk of CVD, according to the researchers. (Note: Another study by Garg et al. presented at the 2019 ACR/ARP Annual Meeting showed that severe renal arteriosclerosis at LN diagnosis increased the risk of CVD tenfold compared with patients without severe renal arteriosclerosis. The study also found a correlation between advanced chronic kidney disease and increased cardiovascular risk in patients with LN, with patients with stage 3 or greater chronic kidney disease at a fivefold increased risk of CVD.2) To date, however, biomarkers or tests to predict future CVD are lacking, limiting the ability to identify early who may develop CVD.
Using renal arteriosclerosis as a biomarker or predictor of CVD risk in LN patients is supported by previous studies showing renal arteriosclerosis predicts CVD in other glomerulonephritis diseases, such as IgA nephropathy as well as in renal transplant patients.3-5 Until now, however, no studies have shown a positive association between the presence of renal arteriosclerosis and CVD occurrence in LN patients.
One reason may be the lack of emphasis in the current International Society of Nephrology/Renal Pathology Society (ISN/RPS) guidelines on universal reporting using standard criteria for renal arterial changes, according to Shivani Garg, MD, MS, assistant professor of medicine, Division of Rheumatology, University of Wisconsin, Madison.
The study bore this out. Garg and colleagues also looked at whether renal arteriosclerosis and its severity were under-reported in pathology reports of LN patients and found they were overlooked or missed in >50% of the pathology reports reviewed.
“Clinicians urgently need early predictors of CVD in LN patients to prevent related morbidity and mortality,” state the authors in the study. “Renal arteriosclerosis on pathology reports could be an early predictor of CVD in LN patients.”
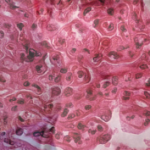
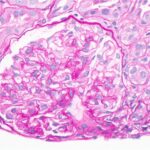
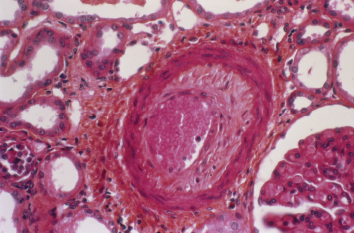
Prevalence of Blood Vessel Disease

Dr. Garg
To test their hypothesis that renal arteriosclerosis would occur at a younger age in patients with LN compared with healthy controls, Garg and colleagues reviewed data on 189 LN patients who underwent native renal biopsy between 1994 and 2017 at the University of Wisconsin Hospital and Clinics. Patients ranged in age from 2–79 years (median age of 25 years), and the majority were female (78%) and white (73%).
Biopsy reports showed that 41% had renal arterial changes (31% had renal arteriosclerosis and 12% had hyaline arteriolosclerosis), and that renal arteriosclerosis increased with age. Overall, one in three patients 60–69 years old had moderate-to-severe sclerosis.
When compared with healthy controls, the onset of any arteriosclerosis in LN patients occurred two decades earlier. Patients with LN aged 30–39 years had comparable renal arteriosclerosis to healthy kidney donors aged 50–59 years (41% vs. 44%), and LN patients aged 40–49 years had comparable renal arteriosclerosis to controls 60–69 years (52% vs. 51%). Patients with LN aged 60-69 years had a fivefold higher prevalence of moderately severe arteriosclerosis compared with age-matched healthy donors (33% vs. 6%).
The study found age and LN chronicity were independent predictors of renal arteriosclerosis. Patients with LN older than 30 years had threefold higher odds of developing renal arteriosclerosis than patients younger than 30 (odds ratio [OR] 3.3; confidence interval [CI], 1.3, 9.1; P=0.02), and patients with chronic LN had a fourfold greater odds (OR 4.0; CI, 1.5, 11.6; P=0.01)
Commenting on these findings, Ram Raj Singh, MD, professor of medicine, rheumatology, and pathology at the David Geffen School of Medicine, University of California, Los Angeles, emphasizes the need for more research to better understand why some patients with LN develop renal arteriosclerosis. He also notes the need for additional research given the small numbers in the study and the predominance of white patients.
“Because nonwhite patients generally have more severe lupus nephritis and poorer outcomes from lupus, future studies involving diverse patient populations would be needed to further realize the importance of this research,” he says.
The authors note the lack of diversity as a study limitation, and according to Dr. Garg, future research will focus on diverse cohorts and the correlation between renal arteriosclerosis and CVD occurrence.