The carbohydrate content of a preparation is particularly important for patients with diabetes mellitus, as glucose-containing products may acutely affect glycemic control. In addition, a disproportionate share of renal adverse events is associated with sucrose-containing products.3,4 Sugar and sodium content also both affect osmolality, and increased osmolality may be of concern in patients with a history of renal dysfunction, elevated blood pressure or heart failure. Lyophilized products needing reconstitution also tend to have higher osmolality than liquid formulations of IVIg. Patients who cannot tolerate increased osmolality may also not tolerate a high volume load, mostly determined by IVIg dose and product concentration. Lastly, all IVIg preparations contain a small amount of IgA, and patients with IgA deficiency may be at risk of anaphylaxis because of their production of IgE anti-IgA antibodies, particularly with prior exposure to blood products containing IgA. Anaphylaxis is rare, however, and most patients with IgA deficiency tolerate products with low IgA content.4
Safety and Infusion-Related Adverse Events
A majority of the side effects associated with IVIg are minor, self-limited, and typically related to the infusion rate (see Table 2). The most commonly reported side effects are headache, nausea, myalgia, arthralgias, urticaria, low-grade fever, chest discomfort, and tachycardia. Milder reactions can often be reduced or prevented by decreasing the speed of the infusion or temporarily discontinuing the infusion and restarting at a lower rate after symptoms resolve. If a patient continues to experience adverse reactions with treatment, a trial of premedications including antihistamines, analgesics, and rarely corticosteroids is reasonable. Switching to a different brand of product is also worth considering.3
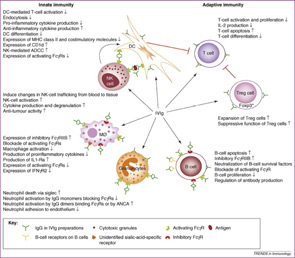
Major side effects of IVIg therapy include aseptic meningitis, acute renal failure, thrombotic complications, and, rarely, anaphylaxis. Patients with a history of migraine headaches in particular have a predisposition toward aseptic meningitis. Prehydration, slower infusion rates, and premedication with analgesics and a migraine-specific medication such as sumatriptan may help prevent this complication. Acute renal failure is thought to be due to an osmotic nephrosis leading to acute tubular necrosis. As previously mentioned, the literature estimates that 90% of cases of acute renal failure are associated with the infusion of sucrose-containing preparations of IVIg. A significant number of patients developing this complication also have other comorbid conditions, including baseline renal insufficiency, hypertension, and diabetes mellitus. Thrombotic events are likely related to increases in plasma viscosity above the physiologyic range of 285–295 mOsmol/kg. Myocardial infarction and stroke are rare and potentially fatal side effects that may be due to increases in serum viscosity. Lower-concentration solutions and slower speeds of infusion may decrease the potential for thrombotic complications. For high doses of IVIg, however, lower concentrations may demand larger volumes of fluid and providers should be careful of volume overload.3,4