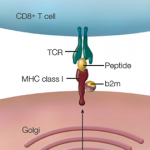
Honestly, I’m not sure why I bothered to listen.
I was in my kitchen, early on a Saturday morning, with the radio keeping me company. It started with the news and the usual litany of tragedies, both major and minor, that comprise such programs. The news gradually faded into other programming, which I can no longer recall; honestly, I was using the radio for background noise, like elevator music for the tone deaf.
Then it hit me: I was listening to a story on sperm donation.
I normally would have just turned off the radio—I’m not a prude, I just tend to avoid listening to stories about medical topics. It’s too much like work. Worse, it is difficult to turn the doctor part of my brain off, so I create a running critique, as if the reporter were a medical student in one of my classes, and I was grading his report.
But my hands were full, and I was distracted, so the story gradually crept into the outer boundaries of my conscience, like an earworm or the latest summer hit, until I was hooked.
He started to donate when he was in college. Outside the pocket change he earned, he thought of it as a civic duty. To each, according to their need; from each, according to their ability.
Decades later, he started to learn how successful his contributions were. At last count, he has met 14 of his progeny. Men are apparently more reluctant to reach out than women, so he is expecting a second wave of children to emerge at some point.
He notes there was no instantaneous connection when he met his progeny. Of course, there were physical similarities; for example, he noticed that many of his offspring had the same flecks of color in their eyes. More surprising to me was another observation: They could all sing.
The father relates a story of how he met his daughter: Not knowing anything about her, he decided to take her to a karaoke bar, which he thought represented neutral territory. While he was singing, he noticed his daughter starting to cry. She later explained that she was raised in a tone-deaf family. Her ability and love of singing had made her feel out of place her entire life.
At first, this doesn’t seem any more remarkable than passing along the color of his eyes. It seems to make sense that the ability to sing might also be genetic. But take a moment to think about how complex this trait must be—the ability to hear a melody, internalize the notes and rhythm, and then reproduce both, must be as complicated as learning a language. That said, everyone speaks some language; not everyone can sing.
Siblings & Other Forms of Torture
“Only if you have an older brother who tortured you when you were a kid,” is my standard reply.