arka38 / SHUTTERSTOCK.COM
SAN DIEGO—Recent research tells us more about giant cell arteritis (GCA) to help rheumatologists more accurately diagnose and effectively treat patients with this type of vasculitis. On Nov. 6 at the ACR/ARHP Annual Meeting, three experts explored the latest findings on GCA pathogenesis, diagnostic approaches, imaging modalities and growing treatment options.
GCA: What’s Really Happening?
GCA is a granulomatous, large-vessel vasculitis that usually affects patients after 50, and women more often than men. GCA may affect the aorta and the smaller, second to fifth aortic branches, particularly the temporal artery, said Cornelia Weyand, MD, PhD, professor of medicine at Stanford University’s Vasculitis Research Center.
Immune privilege usually spares the aorta and its branches from inflammatory attacks, but in GCA, that pact is broken, she said. Patients may have aortic aneurysm, stenosis or occlusion in the aortic branches, tissue ischemia and constitutional symptoms that muscle pain, or polymyalgia rheumatica, as well as malaise, anemia, thrombocytosis, failure to thrive and, sometimes, fever.
GCA has both vascular and extravascular components. In the extravascular phase, innate immune cells like monocytes and neutrophils are triggered, and innate, pro-inflammatory cytokines are released. There is an induction of hepatic acute phase proteins, such as C-reactive protein, mannose-binding protein, ferritin and others. This may result in an inflammatory amplification loop.
“We need to understand the role of hepatic acute phase proteins in GCA and polymyalgia rheumatica, and we need to know what they do in the disease process. Right now, we do not know,” Dr. Weyand said.
Unanswered Questions
Tocilizumab, an IL-6 inhibitor, is approved by the U.S. Food and Drug Administration to treat GCA, but unanswered questions, including whether biomarkers for disease activity are lost after therapy blocks IL-6 and if the tissue-protective functions of acute phase proteins are also blocked by treatment, are important for GCA patients, said Dr. Weyand.
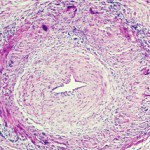
In GCA, T cells, dendritic cells and macrophages form granulomatous lesions inside the vessel walls. CT angiography and magnetic-resonance angiography (MRA) help reveal the degree of disease and which arteries are involved. Disease tends to go into the aortic branches in a symmetrical pattern. As occlusion occurs, blocking off blood flow, blood tends to be rerouted to other vessels. Patients form aneurysms when disease leads to vessel destruction in the aorta, requiring surgical repair.
“We know that the disease leads to dissection, which we can rapidly diagnose. We know that when it dissects, it does so between the adventitia and the media, and forms a dissection lesion,” which can be fatal without intervention, Dr. Weyand showed. “So how does this vasculitic lesion come about? And how is this lesion connected to the extravascular component of disease? The GCA lesion has two major pathomechanisms. It has intimal hyperplasia and intimal myofibroblast proliferation that leads to the occlusion of the lumen. And it has neo-angiogenesis that sits mostly in the adventitia, and that’s what we capture when we image these patients.”
Inflammatory infiltrates in the media and adventitia have different composition. T cells are dominant in these lesions, and these T cells may produce IFN-γ, IL-17, IL-21, IL-9, IL-2 and IL-22, a highly diverse effector cell population that suggests multiple antigens are driving disease, “because so many different inflammatory cells are driving the lesion,” said Dr. Weyand. “What the lesion looks like from an immunological perspective is like it is unopposed, or to use a more modern term, like the immune response is ‘unleashed.’ So the question is how can this unleashed immune response get into a site that is normally protected by immune privilege—a site that nature had determined it does not want to have any inflammatory activity for any price?”
Research findings show three failures in this immune privilege in GCA: Patients have increases in CD4 T cells, a leakiness in the aorta’s access portal that would normally keep out inflammatory cells and defective immune checkpoints.
In their lymphoid tissues, GCA patients have low levels of CD8 T-regulatory cells with a surface marker called NADPH oxidase. Normally, macrophages use this oxidase to form exosomes that infiltrate CD4 cells and stop their function, killing bacteria. Immune dysregulation in GCA causes these patients to have more active CD4 T cells, she said.1
Inflammatory cells enter the aorta through the vasa vasoral networks of the adventitia. Using biopsy data, she and her colleagues found two signals of gene expression in the inflamed arteries of GCA patients: Jagged-1 and NOTCH-1.2 Jagged-1 is the ligand and NOTCH-1 is the receptor, she said. Through high-resolution imaging, they found that Jagged-1 is expressed on the vasa vasorum structures, and the ligand is expressed on the cell surface. With flow cytometry, they also found that the receptor sits on the circulating CD4 T cells of GCA patients.
“We see that patients with GCA have a population of NOTCH-positive cells. If we look in these cells, they have a signal for a protein called HES, which is NOTCH-signaling dependent. Every one of these NOTCH cells is transmitting that signal,” she said. They also found that GCA patients have increased vascular endothelial growth factor (VEGF) that upregulates Jagged-1 on the microvascular cells. Through studies on specially bred mice, the researchers confirmed that both Jagged-1 and NOTCH-1 signaling are VEGF dependent. VEGF increases interferon production in CD3 T cells found in GCA lesions, she said. VEGF may be blocked with axitinib.
Tocilizumab may help GCA patients lower their overall steroid dose & decrease relapse risk.
Checkpoint Breakdown
As in cancer, immune checkpoint function is important in GCA pathogenesis. Tumors evade immune systems by producing negative, inhibitory ligands that stop T cells sent to kill those tumors. In GCA, patients have an immune checkpoint deficiency, said Dr. Weyand. They have low levels of the inhibitory ligand PD-L1 in their inflamed temporal arteries and high levels of the receptor, PD-1.
“This is a clear failure of that immune checkpoint,” she said. Erythrocyte sedimentation rate testing can show that PD-L1 expression on dendritic cells inversely correlates with inflammatory markers in GCA patients. When researchers treated bio-engineered mice with a checkpoint inhibitor to block PD-L1, “we see that it breaks the immune tolerance of the vessel wall.3 If we break the checkpoint with this treatment, we get vigorous GCA disease in these arteries. Checkpoint inhibition also accelerates inflammatory cytokine production.” Treatment raises PD-1 levels, increases pro-inflammatory transcription factors, and enhances IFN-γ, IL-17A and IL-21, she said. It also unleashes T cell immunity, thickens the intima and dramatically increases microvascular angiogenesis in the adventitia.
Tocilizumab, an IL-6 inhibitor, is approved by the U.S. Food & Drug Administration to treat GCA, but unanswered questions, including whether biomarkers for disease activity are lost after therapy blocks IL-6 & if the tissue-protective functions of acute phase proteins are also blocked by treatment, are important for GCA patients, said Dr. Weyand.
Diagnosis & Therapy
To diagnose GCA, rheumatologists typically evaluate the temporal artery, especially in patients with cranial manifestations, said Tanaz A. Kermani, MD, MS, FACP, assistant professor of medicine and director of the Vasculitis Program at the University of California, Los Angeles. Biopsy may detect disease symptoms even weeks after treatment is initiated, but it is invasive, she said. High-resolution magnetic resonance imaging (MRI) of scalp arteries shows some promise as an alternative non-invasive modality for initial GCA diagnosis, and ultr

Dr. Kermani
asound may also be helpful, but the sensitivity of both MRI and ultrasound diminish after patients start treatment.4
A new approach being studied is the fast-track clinic, where rheumatologists promptly evaluate any patients with suspected GCA, including use of ultrasound.5 If GCA is indicated, patients begin treatment right away, said Dr. Kermani. Data from these clinics has shown a dramatic decrease in vision loss, but more prospective data is needed on this approach, she said.
In evaluation, rheumatologists may overlook GCA’s possible extracranial manifestations, such as large artery stenosis or aortic involvement, said Dr. Kermani. Physical examination has poor sensitivity in diagnosing large-vessel vasculitis, so angiography or positron emission tomography (PET) may be used to find lesions in these vessels.6,7
Careful evaluation is important, because a significant portion of GCA patients may develop large-vessel manifestations, especially aortic aneurysms even 10 years after diagnosis.8
“This is an important implication for our patients. They may no longer be following up with rheumatology because they may be off treatment, but this is a late complication of the disease,” she said.
Patients with upper-extremity limb claudication may be younger than others with GCA, who have more typical cranial manifestations.9 Their diagnosis may be delayed because they lack the more common signs of GCA, said Dr. Kermani. “Maybe physicians don’t recognize it or think about it early enough. Inflammation may be in different segments, so they tend to have less vision loss,” she said. These GCA patients may have a negative temporal artery biopsy. “So if you suspect this diagnosis, large-vessel imaging is really the way to proceed in terms of your diagnostic evaluation.”
Although GCA patients should be screened for aortic aneurysms, a late-stage complication, there is no consensus on the type of imaging modality or frequency and timing of screening in these patients, she said. GCA patients are also at increased risk for vertebrobasilar stroke, myocardial infarction and venous thromboembolism, so GCA screening may be considered in older patients who present with these vascular events.10
Glucocorticoids have been the mainstay of GCA therapy in the past, and “prompt treatment in anyone with suspected GCA is essential to prevent vision loss. In patients where vision loss does occur, prompt institution of glucocorticoids can prevent contralateral eye involvement or progression of their visual deficit,” said Dr. Kermani. After a starting dose of 40–60 mg/day for one month, if symptoms resolve and biomarkers normalize, patients may taper their glucocorticoid dose. Patients with vision loss or ischemic complications such as limb claudication may need pulse steroids, she added. Aspirin is not routinely recommended.
Tocilizumab may help GCA patients lower their overall steroid dose and decrease relapse risk, according to the results of a Phase III, randomized clinical trial.11 Other potential therapies now in clinical trials include abatacept and ustekinumab, but these are not yet approved for GCA, she said.12,13
Imaging Strengths & Limitations
Rheumatologists must select the right imaging modality at different points in the GCA disease process, said Peter C. Grayson, MD, MSc, Principal Investigator of the translational research program in vasculitis at the National Institutes of Arthritis and Musculoskeletal and Skin Diseases (NIAMS) of the National Institutes of Health (NIH). Ultrasound of extracranial vessels to diagnose GCA has many advantages, he said.14 It is relatively cheap, non-invasive, involves no radiation and can be performed at bedside.
“It’s a dynamic form of imaging. You can see how blood is flowing through the arteries and see signs of turbulence,” said Dr. Grayson. “The other thing you can see in ultrasound is the wall of the artery and the morphology of it. Is this imaging finding telling me that inflammation is going on in this vessel wall? What you will see, particularly in the cross-sectional view, is you can see this hypo-echoic rim around the lumen of the artery, which is what we call the halo sign. This tells me that there is likely this edematous, thickened arterial wall,” a sign of potential arteritis.
Ultrasound and temporal artery biopsy show widely variable rates of specificity and sensitivity for GCA diagnosis in different studies to compare them, Dr. Grayson said. Patients may have a normal ultrasound, but later be diagnosed with GCA. In a 2016 study comparing the two methods, ultrasound showed 81% specificity but 54% sensitivity for diagnosis of GCA, while biopsy showed 100% specificity but only 39% sensitivity.15 Both ultrasonographers reviewing scans and pathologists reviewing biopsy results may disagree on these findings in GCA, he added.
High-resolution scalp MRA can show abnormalities, such as increased wall thickness in temporal arteries, and has 80% agreement with biopsy, but many patients with a normal MRA still go on to be diagnosed with GCA.4 Fluorodeoxyglucose (FDG) PET scans cannot detect temporal artery inflammation, as the resolution of these scanners is not yet good enough for the job, he said. FDG-PET may be used to examine larger arteries like the aorta, along with angiography, said Dr. Grayson.
Both MR- and CT-based angiography platforms are non-invasive and useful for assessing disease activity and monitor GCA patients for silent disease progression. However, it is hard to cover the whole body in one session, specificity is lacking, and cost, radiation and potential adverse effects to the patient are valid concerns.
Weaknesses: It’s hard to cover the whole body in one session. Technical expertise is required for these modalities, he said.
Because it uses glucose, PET may detect metabolic activity in the walls of large arteries that suggest inflammation. PET may be used to categorize the extent of disease and assess disease activity in GCA patients, as well as monitor for silent progression of disease and, possibly, treatment response. However, these tests are hard to obtain, he said. Other limitations include a lack of a gold standard for PET’s use as a diagnostic tool in GCA, as well as cost and the use of radiation. It cannot be used to correctly assess the temporal arteries. While a 2015 meta-analysis of FDG-PET to diagnose GCA showed both high specificity and sensitivity, selection bias may have elevated those numbers, he said.16 At this time, it seems that all available imaging modalities have a role to play in diagnosis and ongoing assessment of GCA patients.
Susan Bernstein is a freelance journalist based in Atlanta.
References
- Wen Z, Shimojima Y, Shirai T, et al. NADPH oxidase deficiency underlies dysfunction of aged CD8+ Tregs. J Clin Invest. 2016;126(5):1953–1967.
- Wen Z, Shen Y, Berry G, et al. The microvascular niche instructs T cells in large-vessel vasculitis via the VEGF-Jagged-1-NOTCH pathway. Sci Transl Med. 2017 Jul;399(9):eaal3322.
- Zhang H, Watanabe R, Berry GJ, et al. Immunoinhibitory checkpoint deficiency in medium and large vessel vasculitis. PNAS. 2017;114(6):E970–E979.
- Rheaume M, Rebello R, Pagnoux C, et al. High-resolution magnetic resonance imaging of scalp arteries for the diagnosis of giant cell arteritis: Results of a prospective cohort study. Arthritis Rheumatol. 2017 Jan;69(1):161–168.
- Patil P, Williams M, Maw WW, et al. Fast track pathway reduces sight loss in giant-cell arteritis: results of a longitudinal observational cohort study. Clin Exp Rheumatol. 2015 Mar–Apr;33:(2 suppl 89):S-103–106.
- Grayson PC, Tomasson G, Cuthbertson D, et al. Association of vascular physical examination findings and arteriographic lesions in large vessel vasculitis. J Rheumatol. 2012 Feb;39(2):303–309.
- Prieto-Gonzalez S, Arguis P, Cid MC. Imaging in systemic vasculitis. Curr Opin Rheumatol. 2015 Jan;27(1):53–62.
- Kermani TA, Warrington KJ, Crowson CS, et al. Large-vessel involvement in giant cell arteritis: A population-based cohort study of the incidence-trends and prognosis. Ann Rheum Dis. 2013 Dec;72(12):1989–1894.
- Muratore F, Kermani TA, Crowson CS, et al. Large-vessel giant cell arteritis: A cohort study. Rheumatology (Oxford). 2015 Mar;54(3):463–470.
- Amiri N, De Vera M, Choi HK, et al. Increased risk of cardiovascular disease in giant cell arteritis: A general population-based study. Rheumatology (Oxford). 2016 Jan;55(1):33–40.
- Stone JH, Tuckwell K, Dimonaco S, et al. Trial of tocilizumab in giant cell arteritis. New Engl J Med. 2017 Jul;377:317–328.
- Langford CA, Cuthbertson D, Ytterberg SR, et al. A randomized, double-blind trial of abatacept (CTLA-4Ig) for the treatment of giant cell arteritis. Arthritis Rheumatol. 2017 Apr;69(4):837–845.
- Conway R, O’Neill L, O’Flynn E, et al. Ustekinumab for the treatment of refractory giant cell arteritis. Ann Rheum Dis. 2016 Aug;75(8):1578–1579.
- Laria A, Lurati A, Scarpellini M. Color duplex ultrasonography findings of temporal arteries in a case of giant cell arteritis: Role in diagnosis and follow-up. Open Access Rheumatol. 2017 Mar 15:9:55–59.
- Luqmani R, Lee E, Singh S, et al. The role of ultrasound compared to biopsy of temporal arteries in the diagnosis and treatment of giant cell arteritis (TABUL): A diagnostic accuracy and cost-effectiveness study. Health Technol Assess. 2016 Nov;20(90):1–238.
- Soussan M, Nicolas P, Schramm C, et al. Management of large-vessel vasculitis with FDG-PET: A systematic literature review and meta-analysis. Medicine (Baltimore). 2015 Apr.94(14):e622.