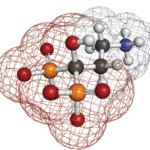
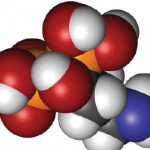
Bisphosphonate Drug Holidays
ajt/shutterstock.com
Drug holidays are common for patients on bisphosphonate therapy. Often, these breaks in treatment are related to known U.S. Food and Drug Administration (FDA) warnings and drug class adverse effects. Currently, data on fracture risk related to drug holidays are limited. In recent research highlighted at the 2017 ACR/ARHP Annual Meeting , investigators conducted a population-based cohort study of long-term bisphosphonate use in women to evaluate the hip fracture rate after a drug holiday.1
Researchers used Medicare data from 2006–2014 to identify women with medical and pharmacy coverage who initiated bisphosphonate therapy and were at least 80% adherent to that therapy for a minimum of three years (baseline) when follow-up began. Researchers calculated crude hip fracture rates for women who continued to receive bisphosphonates and for those who discontinued the treatment. The rates were calculated for time since discontinuing treatment (i.e., drug holiday length), extending up to three years.
The study identified 156,236 women who were highly adherent, long-term users of bisphosphonates. The mean patient age was 78.5 (SD=7.5) years. Alendronate was the most commonly used bisphosphonate, with 72% of patents having used the agent and 52% having exclusively used it. Zoledronic acid was used by 16% of women, with 9% of women having exclusively used this agent. Thirteen percent of women (7,947) subsequently resumed bisphosphonate therapy.
During a median follow-up of two years, 40% of women discontinued using bisphosphonate treatment for at least six months (n=62,676). Thirteen percent of these women (n=7,947) restarted a bisphosphonate at a later date.
During follow-up, 3,745 hip fractures occurred. Current bisphosphonate users had the lowest rate of hip fracture (9.6% crude incidence per 1,000 person-years). Patients who had a longer drug holiday experienced higher rates of hip fracture. The longer the drug holiday, the higher the rate of hip fracture. Women who had a one- to two-year drug holiday had a 13.3% crude incidence of hip fracture per 1,000 person-years. Women who had a two- to four-year drug holiday had a 15.7% crude incidence of hip fracture per 1,000 person-years. Overall, 11% (n=16,904) of patients died during the study, with details not provided.
Golimumab Receives 2 More FDA Approvals
In late October, the FDA approved golimumab to treat adults with active psoriatic arthritis (PsA) or active ankylosing spondylitis (AS). The medication was previously approved to treat moderate to severe active rheumatoid arthritis. Golimumab, a fully human anti-TNF-alpha monoclonal antibody, targets soluble and transmembrane bioactive forms of human TNF-alpha, which cause inflammation when overproduced.2