 BALTIMORE—Many rheumatologists know that vasculitis can rapidly lead to morbidity and mortality for afflicted patients. Thus, understanding the advances in care for vasculitis is key to preventing patient suffering and saving lives.
BALTIMORE—Many rheumatologists know that vasculitis can rapidly lead to morbidity and mortality for afflicted patients. Thus, understanding the advances in care for vasculitis is key to preventing patient suffering and saving lives.
At the 18th Annual Advances in the Diagnosis & Treatment of the Rheumatic Diseases, held May 13–14 at Johns Hopkins School of Medicine, Baltimore, Brendan Antiochos, MD, assistant professor of medicine, Division of Rheumatology, Johns Hopkins School of Medicine, and assistant director of the Johns Hopkins Vasculitis Center, discussed important topics in vasculitis. He addressed new medications for anti-neutrophil cytoplasmic antibody (ANCA) associated vasculitis and giant cell arteritis (GCA), tapering of glucocorticoids in patients with vasculitis and issues related to vaccination against SARS-CoV-2 in patients with vasculitis, particularly those receiving B cell-depleting therapy.
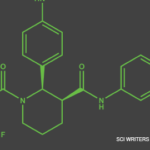
Avacopan
Dr. Antiochos began by discussing the ADVOCATE trial, a large, international, randomized clinical trial comparing avacopan (a C5a receptor inhibitor) with prednisone in patients with ANCA-associated vasculitis who were receiving either cyclophosphamide plus azathioprine, or rituximab.1
Dr. Antiochos explained that the C5a receptor can be found on myeloid cells and that activation of this receptor helps recruit these cells to sites of injury or inflammation. Avacopan is designed to block the C5a receptor and prevent such recruitment without affecting the formation of the membrane attack complex, which is a product of the complement system and helps protect against certain pathogens.
In 2014, researchers tested a mouse model of necrotizing and crescentic glomerulonephritis mediated by anti-myeloperoxidase antibodies, as would be seen in ANCA-associated vasculitis. They found oral administration of avacopan—called CCX168 at that time—prevented anti-myeloperoxidase-induced necrotizing and crescentic glomerulonephritis in mice expressing human C5aR/CD88.2
In the ADVOCATE trial, patients were randomized to receive either 30 mg of avacopan twice daily or an oral prednisone taper, with background therapy of either cyclophosphamide induction followed by azathioprine maintenance or rituximab. The main characteristics of study participants were patients who were newly diagnosed and had seropositive ANCA-associated vasculitis. The majority had renal disease and had been treated with rituximab. The study’s primary end points were clinical remission at week 26, with a Birmingham Vasculitis Activity Score (BVAS) of 0 and no steroids for at least four weeks before week 26, and sustained remission at weeks 26 and 52, with no glucocorticoids for at least four weeks before week 52.
The results: Avacopan proved noninferior to the oral prednisone taper for clinical remission at week 26 and was superior to the oral prednisone taper for sustained remission at week 52. Roughly 10% of the avacopan patients experienced a relapse of disease at week 52 compared with about 20% of those in the oral prednisone taper group. In total, patients in the avacopan group received about one-third less total prednisone than those in the oral prednisone taper group. An additional exciting finding described by Dr. Antiochos was that renal function in patients treated with avacopan continued to improve throughout the study.1