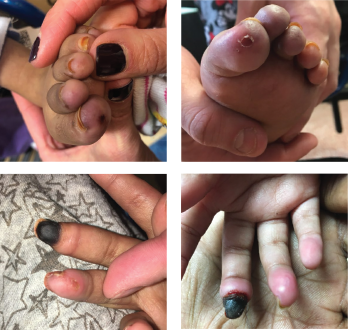
Figures 1A-D: A 5-year-old girl presented with erythema and ulcerations of the finger- and toetips, which had progressed to ischemia of the right, third fingertip.
Janus kinase 1 and 2 inhibitors (jakinibs) have been approved for the treatment of rheumatoid arthritis, psoriatic arthritis and, most recently, juvenile idiopathic arthritis. They have also shown promise in the treatment of interferon (IFN) mediated diseases. The Janus kinase and signal transducer and activator of transcription (JAK/STAT) pathway is the principal signaling pathway for IFN receptors, which are upregulated in IFN-mediated diseases, such as Aicardi-Goutières syndrome.
Here, we present a case of a child who presented with a necrotic digit and was found to have Aicardi-Goutières syndrome, which had previously been misdiagnosed as metachromatic leukodystrophy. She would potentially have benefited from treatment with a jakinib, but was unable to obtain insurance approval for this medication.
Case Presentation
A 5-year-old girl with a diagnosis of metachromatic leukodystrophy, spastic quadriplegia and developmental delay presented with a six-week history of erythematous discoloration and ulcerations of the fingertips and toes (see Figures 1A–B) and ischemia of her right third fingertip (see Figures 1C–D). She was admitted to the hospital for evaluation and treatment of the ischemic digit.
She had no history of fevers or joint swelling, no known trauma and no significant cold exposure to the affected areas. In addition to the findings described on her digits, a physical exam revealed normal vital signs, hypertonia and contractures consistent with her metachromatic leukodystrophy diagnosis, no swollen joints or effusions, and no other skin lesions or rashes. Her respiratory and cardiac exams were also unremarkable.
An evaluation targeting a diagnosis for her ischemic finger was undertaken. The investigation for an underlying hypercoagulable state was negative. She had a prothrombin time test, which revealed a normal international normalized ratio (INR), and her fibrinogen, protein C and S, and antithrombin 3 levels were within normal ranges. Tests for antiphospholipid antibodies and cryoglobulins were negative.
An echocardiogram was obtained to assess for a thromboembolic etiology and did not demonstrate valvular vegetations.
A vasculitis or underlying autoimmune disease with associated vasospasm was considered. A computed tomography (CT) scan of her head, neck, chest, abdomen and pelvis revealed normal vasculature.
Her erythrocyte sedimentation rate (ESR) was 95 mm/hr (reference range [RR] 0–15 mm/hr). Her total IgG was elevated at >2,000 mg/dL (RR: 454–1,360 mg/dL). She had an unremarkable urinalysis and her serum creatinine, C3 and C4 were all within the normal range. Her anti-nuclear antibody (ANA), double-
stranded DNA and anti-neutrophil cytoplasmic antibody (ANCA) were all negative. An extractable nuclear antigen (ENA) antibody panel was also negative.
Two skin biopsies were performed. The first biopsy showed some dermal mucin deposition, which can be seen in chilblain lupus erythematosus, but a repeat biopsy was nonspecific, demonstrating only perivascular inflammation.
At this point, she received a diagnosis of perniosis without any clear underlying autoimmune disease. Her care team took a step back, and in the setting of her neurologic abnormalities, they considered Aicardi-Goutières syndrome as a diagnosis that could elegantly tie together her acute presentation with her past history of spastic quadriplegia and developmental delay.


