Prognostic Value
Dactylitis and enthesitis are both regarded as signs of PsA disease severity. Dactylitis is associated with joint erosions.14 Enthesitis is associated with worse disease and radiographic damage in psoriatic arthritis.9 Depending on location (e.g., the Achilles tendon), dactylitis can cause disability and adversely impact a patient’s life in addition to other PsA manifestations.
Treatment
To date, no studies have been sufficiently powered to investigate the treatment of dactylitis and enthesitis, but several studies have reported them as secondary endpoints. The Group for Research and Assessment of Psoriasis and Psoriatic Arthritis (GRAPPA) and the European League Against Rheumatism (EULAR) recommend corticosteroid injections and conventional synthetic disease-modifying anti-rheumatic drugs (DMARDs) as first-line therapy for dactylitis, and NSAIDs and physiotherapy as first-line therapy for enthesitis. In patients who fail to respond, these steps are followed by biologics (e.g., TNF inhibitors).15,16
NSAIDs work by downregulating PGE2 production, an early mediator of entheseal inflammation. Ultrasonography studies have shown NSAIDs decrease vasodilation and inflammation at entheses.17 Note: Once enthesitis becomes chronic, it is anecdotally harder to treat and less responsive to NSAIDs.
Conventional synthetic DMARDs are generally not efficacious for enthesitis, and switching to a biologic therapy as a next step is reasonable. TNF inhibitors have shown benefit for both axial and peripheral enthesitis in a variety of studies (though they were not designed to look at this as a primary outcome). Infliximab, adalimumab, etanercept, golimumab and certolizumab are all viable options, though head-to-head comparisons have not been made between these drugs for clinical dactylitis and enthesitis.18-22
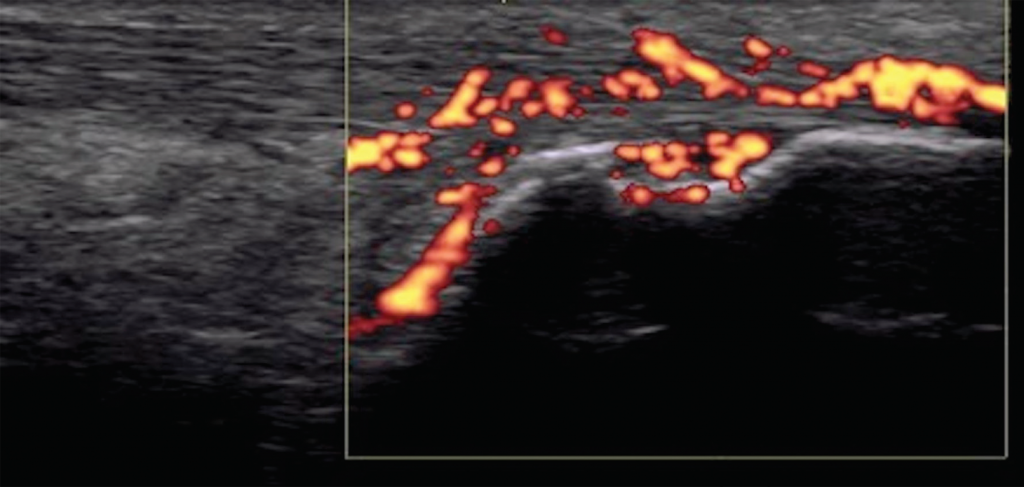
Figure 7. Achilles tendon enthesitis with active Doppler; erosion of calcaneus.
MRI studies of patients with spondyloarthritis (predominantly of the ankylosing spondylitis subset) have shown improvement of peripheral joint, sacroiliac joint and vertebral body peri-entheseal osteitis with TNF inhibitors.23 Secukinumab, ixekizumab (which are anti-IL-17A inhibitors) and ustekinumab (an anti-IL12/23 inhibitor) have also shown efficacy in both clinical dactylitis and enthesitis.24-26
GRAPPA recommends switching to alternate mechanisms of action if TNF inhibitors fail to provide adequate relief. Apremilast, an oral phosphodiesterase 4 inhibitor, has been shown to have some efficacy in axial enthesitis (MASES). Apremilast decreases the production of several cytokines involved in entheseal inflammation (IL-17A, IL-23 and TNF-α), and also decreases neutrophil migration to the entheseal complex. A study of 504 patients showed improvement in axial enthesitis in about half of study participants by 12 months of therapy.27 More studies are needed to determine how effective apremilast is for peripheral enthesitis and for dactylitis.