Scleromyxedema is a primary cutaneous mucinosis characterized by a diffuse and generalized papular skin eruption of mucinous deposits throughout the upper dermis. In addition to dermatologic manifestations, scleromyxedema may involve the cardiopulmonary, gastrointestinal, renal and nervous systems.
Dermato-neuro syndrome (DNS) is a rare, severe neurologic complication of scleromyxedema.1,2 The pathogenesis of DNS is unknown, but circulating cytokines may play a role. With intravenous immunoglobulin (IVIG) maintenance therapy, recurrence is rare.
Coronavirus disease 2019 (COVID‑19), caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), primarily causes respiratory problems; however, central nervous system (CNS) manifestations have also been reported. Neurological complications of COVID-19 include headache, encephalopathy, hyposmia and dysgeusia.3 Multiple mechanisms contribute to neurologic complications of COVID-19, including coagulopathy, inflammatory vasculopathy and cytokine release syndrome.
We present the case of a patient with well-controlled scleromyxedema on maintenance IVIG therapy who had DNS recurrence secondary to COVID-19, illustrating the potential role of SARS-CoV-2 in immune dysfunction and the pathogenesis of DNS.
Case Presentation
A 58-year-old woman with well-controlled scleromyxedema and a prior episode of DNS, who was being treated with levetiracetam and IVIG maintenance therapy without interruption in therapy, presented with encephalopathy and a one-week history of shortness of breath and upper respiratory symptoms. The examination was remarkable for disorientation, generalized decreased motor strength and diffusely tight and hypertrophic skin compared with baseline. She had intermittent rhythmic right-arm shaking episodes that generalized into prolonged tonic-clonic convulsions with associated urinary incontinence.
Continuous electroencephalogram showed non-convulsive status epilepticus (NCSE) that progressed to super-refractory NCSE (see Figure 1). The patient was intubated and treated with levetiracetam (60 mg/kg intravenous load, followed by 2,000 mg twice daily), fosphenytoin (20 mg/kg intravenous load, followed by 200 mg twice daily), lacosamide (300 mg intravenous load, followed by 200 mg twice daily), phenobarbital (15 mg/kg intravenous load, followed by 100 mg twice daily), and midazolam (0.2 mg/kg intravenously administered, followed by continuous infusion with repeat loading dose every five minutes to a maximum total dose of 1 mg/kg) and pentobarbital infusion (15 mg/kg administered at a rate of ≤50 mg/minute, followed by 1 mg/kg/hour continuous infusion to achieve burst suppression for 48 hours, followed by gradual withdrawal within 24 hours to prevent recurrent status epilepticus).
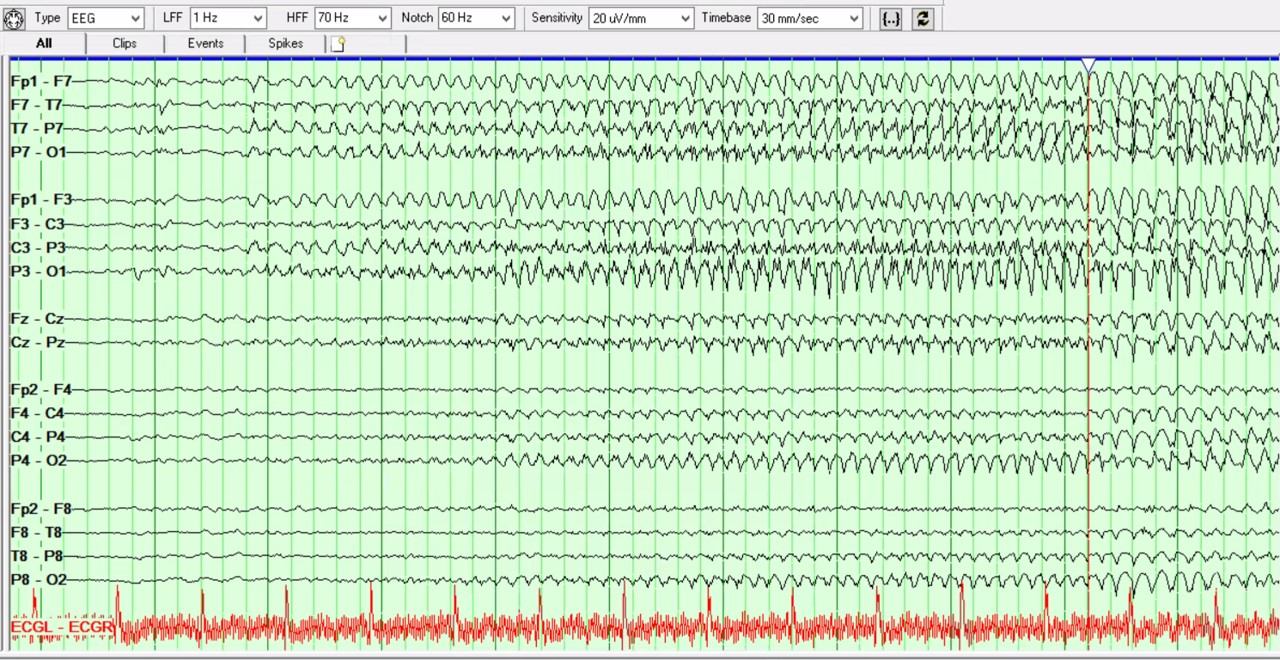 This bipolar montage shows a seizure arising from burst activity, with spikes over the
This bipolar montage shows a seizure arising from burst activity, with spikes over the
left hemisphere, evolving into the left more than the right hemisphere, with bilateral,
rhythmic, sharply contoured theta activity. The EEG shows further rhythmic, spike/