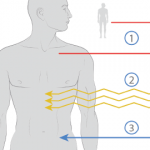
To obtain cells for autologous HSCT before conditioning, stem cells are mobilized from the marrow to the peripheral blood, which permits stem-cell collection without general anesthesia or bone-marrow harvest. This mobilization is achieved by various protocols, many of which involve granulocyte colony-stimulating factor (G-CSF) or cyclophosphamide, a drug that is myelosuppressive and immunosuppressive per se but leads to a “rebound” mobilization of stem cells. The autologous hematopoetic stem cells are then collected through leukapheresis. In a further step, the hematopoetic stem cells expressing CD34+ as a stem-cell marker can be purified through positive selection using CD34 + sorting or negative selection of T or B cells using lineage-specific surface markers. This procedure leads to a more profound T cell depletion of the autograft (up to a CD3+ cell content lower than one T cell per 105 cells in the graft).
Stem-cell mobilization is an immunosuppressive process, especially if cyclophosphamide is used. More profound immunosuppression is subsequently achieved through conditioning before HSCT. Conditioning means that an immunosuppressive regimen causes an at least transient bone marrow insufficiency, leading to a pancytopenia. The aim of conditioning is to eliminate to the greatest extent possible autoreactive cells, mostly T or B cells. The dose-limiting factor for such an intense immunosuppression is the hematotoxicity. However, hematotoxicity is reduced by retransfusion of autologous hematopoetic stem cells after conditioning. In any case, a transient pancytopenia occurs with a nadir around day 10 after HSCT. Transfusion of red cells or platelets may be necessary. Prophylactic antibiotic, antifungal, and antiviral therapy are given depending on the intensity of leuco- or lymphopenia. For conditioning chemotherapy, cyclophosphamide, busulfan or radiotherapy, or antibodies against components of the immune system have been used alone or in combination.
Allogeneic HSCT is associated with GvHD that carries significant morbidity and mortality. Theoretically, allogeneic HSCT offers the replacement of an autoreactive immune system with one that is putatively normal, but, in practice, this has yet to be proven. In fact, there are case reports of allogeneic HSCT being performed in patients with rheumatoid arthritis (RA) suffering from drug-induced aplastic anemia and, despite four years of remission, relapse occurred with all immune competent cells being of donor origin. On the other hand, similar cases have had up to 20 years clinical and serological remission.
Although hematopoetic reconstitution occurs within two weeks after HSCT, complete regeneration of the adaptive immune system, in contrast, is delayed up to years. It is thought that as the immune system returns in the presence of the inciting autoantigen, tolerance occurs.