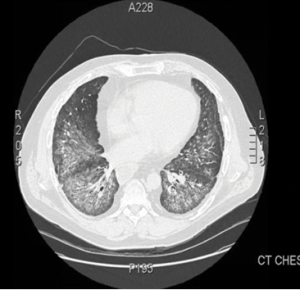
Figure 1.
This computed tomography of the chest shows bilateral, diffuse, ground-glass opacities and nodular opacities with intralobular septal thickening.
Antisynthetase syndrome (AS) is strongly associated with the presence of antibodies to aminoacyl-transfer RNA (tRNA) synthetases (ARSs) that are implicated in the pathogenesis of myositis and interstitial lung disease (ILD).
Antibodies against eight antisynthetases have been identified and are detected in 16–26% of patients with idiopathic inflammatory myopathies (IIM).1 Serum assays for five of these antibodies are commercially available: anti-histidyl (Jo-1), anti-threonyl (PL-7), anti-alanyl (PL-12), anti-isoleucyl (OJ) and anti-glycl (EJ). Anti-Jo-1 antibodies are seen more commonly in patients with polymyositis and dermatomyositis compared with the others that are found less frequently.2 These antibodies usually target the enzymatic component of tRNA synthetases; only anti-PL-12 recognizes tRNA directly.
Few articles in the literature describe the outcome of patients with anti-PL-12 AS. We present a case of anti-PL-12 AS, along with discussion of the outcome, functional course, complications and mortality in AS patients presenting with this rare antibody, along with a brief review of myositis-related antibodies.
Case
In early 2009, a 49-year-old Caucasian male presented to the outpatient rheumatology clinic with complaints of progressive shortness of breath. He had a history of undifferentiated connective tissue disease, sclerodactyly and Raynaud’s symptoms. A review of systems was positive for diffuse joint pains, intermittent fevers, hyperkeratotic skin lesions of both hands, esophageal dysmotility and decreased motility in his gut. His medical history was also significant for splenic vein thrombosis and deep venous thrombosis, for which he was on chronic anticoagulation.
The physical examination revealed decreased bilateral breath sounds, puffy fingers, hyperkeratotic skin over the palmar surface of both hands, no clinical evidence of synovitis and normal muscle strength.
Laboratory analysis revealed mild inflammation with C-reactive protein (CRP) of 15 mg/L, normal creatine phosphokinase (CPK) and normal liver function tests. Antinuclear antibodies (ANA) were negative, and no anti-cytoplasmic staining was detected on indirect immunofluorescence. Rheumatoid factor and anticyclic citrullinated protein antibodies were negative.
An additional workup showed negative anti-neutrophil cytoplasmic antibodies, negative anti-SSA antibodies, and negative PM-Scl antibodies. Pulmonary function tests revealed mild restrictive ventilator defect with severe reduction in diffusion capacity and forced vital capacity (FVC). Forced expiratory volume in 1 second (FEV 1) was 75%, and carbon monoxide diffusion capacity (DLCO) was 36%. High-resolution computed tomography (CT) of the chest demonstrated bilateral pulmonary infiltrates with alveolitis.
The patient was monitored regularly with clinical, laboratory, radiological and lung function evaluations for four years. He developed progression of ILD with declining pulmonary function tests. High-resolution CT imaging showed bilateral pulmonary infiltrates and alveolitis with diffuse, bilateral, ground-glass opacities and nodular infiltrates with intraseptal wall thickening compatible with nonspecific interstitial pneumonia (see Figure 1).

