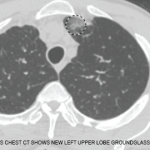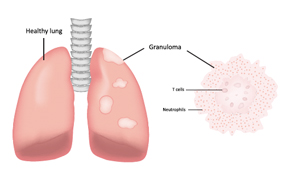
One severe life-threatening vasculitis is granulomatosis with polyangiitis.
joshya/shutterstock.com
SAN FRANCISCO—The heterogeneity of systemic vasculitis, a set of diseases characterized by inflammation of blood vessel walls, presents rheumatologists with diagnostic and treatment challenges, said Sharon A. Chung, MD, MAS, director of the University of California, San Francisco Vasculitis Clinic, at the California Rheumatology Alliance 2016 Scientific & Medical Meeting in May.

Dr. Chung
She outlined emerging therapy strategies for three types of vasculitis: giant cell arteritis, granulomatosis with polyangiitis and Churg-Strauss syndrome. Therapies under study include biological agents, such as rituximab, abatacept, tocilizumab and mepolizumab, which are being used or studied for the treatment of the other autoimmune disorders highlighted in San Francisco.
“There has been significant progress in the past 10 years in the treatment of systemic vasculitis with the advent of biologic therapies, and with multiple studies underway to help expand therapeutic options available to patients,” she said. “In the future, we hope we’ll have an expanded armamentarium of drugs for these diseases.” Meanwhile, the search continues for the holy grail of vasculitis treatment—targeted therapies that minimize toxicity and offer true steroid-sparing benefits. “In vasculitis, we are taking medications shown to be effective for other rheumatic illnesses and seeing if they can be effective for these diseases—because there might be some common pathologies,” Dr. Chung said.
GCA Preliminary Results
The current first-line therapy for giant cell arteritis (GCA) is prednisone. However, there is substantial clinical need for alternative treatments for patients who have GCA that is refractory to glucocorticoid use, cannot taper prednisone without increased disease activity, or have significant adverse effects from prednisone, Dr. Chung said. She reviewed recent studies of tocilizumab and abatacept, monoclonal antibodies now used to treat rheumatoid arthritis and other inflammatory joint diseases, in the treatment of GCA.
A Phase 2, double-blind, placebo-controlled trial from Switzerland reported in The Lancet in May, offered results that Dr. Chung called “astounding.”1 In this study, 30 patients with new-onset or relapsing GCA were randomized to receive either tocilizumab with prednisone or prednisone alone. Eighty-five percent of patients treated with tocilizumab were in complete remission at 12 weeks and at 52 weeks, compared with 40% and 20% of controls, respectively. Patients treated with tocilizumab took significantly less prednisone compared with those taking placebo. However, adverse events, including gastro-intestinal complications and eye infections, also occurred from using tocilizumab.
“This drug potentially could be first-line therapy along with prednisone for GCA, but we’re not there yet,” she said, because the results are from such a small sample. “We are awaiting results from the larger-scale GiACTA study, expected in 2018. But the prospect on the horizon is that tocilizumab could represent a long-needed complement or potential alternative to prednisone for GCA,” she said.