This is Part One of a two-part series. Part Two, on rheumatoid factor and anti-cyclic-citrullinated peptide, will appear in the April issue of The Rheumatologist.
Laboratory testing is an essential element in the diagnosis and management of patients with rheumatic disease. This article focuses on a diverse array of serological markers that can provide unique information on the status of the patient’s immune system that is important in clinical evaluation as well as scientific inquiry. These tests help in the diagnosis of a particular disease and, importantly, they may help monitor disease activity. Indeed, immunological testing represents one of the bedrocks of rheumatology and is a distinguishing feature of our specialty.
While there are many available tests, the approach to serology follows the traditional approach of any laboratory study. Critical in the interpretation of any serological test is determining its sensitivity (e.g., that proportion of patients with the target disorder who have a positive test), specificity (e.g., that proportion of patients who are free of the target disorder and have negative or normal test results), and positive and negative predictive values, which calculate the likelihood that disease is present or absent based on test results using the test’s sensitivity, specificity, and the probability of disease before the test is performed (pretest probability). This review will cover data on antinuclear antibodies (ANAs).
Antinuclear Antibodies
ANAs are the signature autoantibodies of the rheumatic disease and are tested in many clinical scenarios.1-3 These antibodies are usually detected by immunofluorescent (IF) techniques, utilizing Hep-2 cell lines, and specific antinuclear antibodies are detected by solid-phase immunoassays—for instance, an ELISA. The IF ANA is generally screened at a dilution (e.g., titer) of 1:40, and, if positive, serial dilutions are carried out until a dilution is negative. Most labs titer to 1:1280, but some go higher. It is not clear whether titering higher is clinically useful, because the titer of an ANA usually does not correlate with clinical activity.
The IF ANA test is especially useful as an initial screen for patients suspected of having systemic lupus erythematosus (SLE). ANA testing is also useful for the evaluation of patients with other suspected rheumatic conditions including Sjögren’s syndrome, mixed connective tissue disease (MCTD), drug-induced lupus erythematosus, dermato/polymyositis, scleroderma, adult and juvenile arthritis, and autoimmune hepatitis. However, the presence of an ANA does not signify the presence of illness, because these antibodies may also be found in otherwise normal individuals. As shown in Table 1 (see p. 17), the sensitivity of the ANA for a particular autoimmune disease can vary widely.
Other well-recognized conditions that are occasionally associated with a positive ANA include chronic infectious diseases, such as mononucleosis, subacute bacterial endocarditis, and tuberculosis; some lymphoproliferative diseases; and up to 90% of patients taking certain medications, especially procainamide and hydralazine. However, most of the patients who take these medications and develop a positive ANA test do not develop clinical drug-induced lupus.
False-positive ANAs (i.e., ANAs in the absence of autoimmune disease or other diseases) are commonly found in normal women, elderly individuals, and first-degree relatives of patients with ANA-positive autoimmune diseases (typically in low titer). There are several important points about ANA that should be considered in the clinical setting.
TABLE 1: Sensitivity of the ANA in Autoimmune and Nonrheumatic Disease
Autoimmune Disease
- SLE: 95–100%
- Scleroderma: 60–80%
- Mixed connective tissue disease: 100%
- Polymyositis/dermatomyositis: 61%
- Rheumatoid arthritis: 52%
- Rheumatoid vasculitis: 30–50%
- Sjögren’s syndrome: 40–70%
- Drug-induced lupus: 100%
- Discoid lupus: 15%
- Pauciarticular juvenile chronic arthritis: 71%
Nonrheumatic Disease
- Hashimoto’s thyroiditis: 46%
- Graves’ disease: 50%
- Autoimmune hepatitis: 100%
- Primary autoimmune cholangitis: 100%
- Primary pulmonary hypertension: 40%
- The presence of high concentrations of antibody (titer >1:640) should make one suspicious that an autoimmune disorder is present. In this scenario, I recommend that sera be then tested for antibodies to dsDNA, Sm, RNP, Ro (SS-A), La (SS-B), and perhaps Scl-70. The presence of antibodies to any of these greatly increases the likelihood that the patient has SLE, MCTD, Sjögren’s, or scleroderma. Some labs will automatically test for these antibodies whenever the screening ANA is positive. However, the presence of these antibodies is not diagnostic of disease. If no initial diagnosis can be made, it is my practice to watch the patient carefully over time for the development of an ANA-associated disease, and see the patient at least twice yearly.
- The combination of low titers of antibody (<1:80) and no or few signs or symptoms of disease portends a much smaller likelihood of an autoimmune disease. As a result, these patients with low ANA titers need to be reevaluated less frequently—yearly unless clinical symptoms evolve to suggest an autoimmune disease.
- A patient with a negative ANA is highly unlikely to have either SLE, MCTD, Sjögren’s, or scleroderma. However, if there is still strong clinical evidence of a systemic autoimmune disorder, one may test for the specific antibodies to double-stranded (ds) DNA, Sm, RNP, Ro, La, or Scl-70, although, in my experience, they are typically negative. Nevertheless, it is prudent to see such patients where there is a high clinical index of suspicion, at least yearly—although more frequently if clinically indicated.
- Antinuclear antibodies produce a wide range of staining patterns (homogenous, diffuse, peripheral, rim, speckled, nucleolar, anticentromere, etc.). The nuclear staining pattern has been recognized to have a relatively low sensitivity and specificity for different autoimmune disorders. The presence of antibodies directed at specific nuclear antigens is usually more useful. (These antibodies include the following: dsDNA, Sm, RNP, Ro, La, or Scl-70.)
Over the last few years, investigators and biotech firms have been developing solid-phase immunoassays to replace the IF ANA test.4-23 The rationale behind this attempt relates to performance characteristics of the IF technique. This test is very labor intensive and is subject to variation due to different interpretations by technicians. Also complicating testing is the fading of the image as it is examined in a fluorescent microscope. Furthermore, the IF technique uses serial dilutions of patient sera, which will give results that may not be linear. Variations in titer by two-fold are common in day-to-day testing on the same sample; four-fold differences are said to be “significant.” By contrast, solid-phase immunoassays are automated and highly reproducible. The results are linear, and the technique is less labor intensive, and thus cheaper to perform.
Because of these considerations, there should be economic savings in employing a solid-phase immunoassay for quantitating an ANA. Thus, in an attempt to develop solid-phase ANA immunoassays, a number of groups have put onto the solid phase whatever antigens are typically assayed in the more specific ANA immunoassays (e.g., DNA, Sm, RNP, Ro/SSA, La/SSB, nucleoprotein, cell extracts, etc.). In published reports, the correlation coefficient between ANA titers and these solid-phase assays is quite good. Thus, many commercial firms have switched their ANAs to these solid-phase immunoassays.4-23 Of concern, however, is the high frequency/percentage of false-negative results in patients with known SLE and related diseases, as well as the continued high frequency of “false positives” (e.g., a positive ANA in someone without SLE) in these studies.15-21 Further work is needed to improve the sensitivity and especially the specificity of these solid-phase immunoassays to ensure that patients with SLE and related diseases are not missed by these solid-phase immunoassays.
Antibodies to DNA
Antibodies to DNA can be primarily divided into those that react with single-stranded (ss) DNA and those recognizing dsDNA.24-26
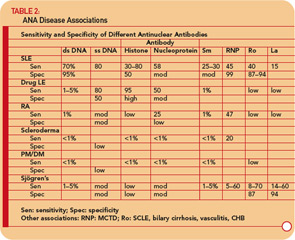
Anti-ssDNA antibodies have been reported in SLE, rheumatoid arthritis, drug-related lupus, healthy relatives of patients with SLE, and, less commonly, in other rheumatic diseases. (See Table 2, p. 17.) Thus, anti-ssDNA has limited usefulness for the diagnosis of SLE or other rheumatic diseases. Anti-ssDNA does not correlate well with disease activity and are therefore not useful for disease management.
Antibodies to dsDNA are most frequently detected by solid-phase immunoassays, but also in some labs by the crithidia IF assay or occasionally the Farr radioimmunoassay. Most hospital and commercial labs do only one. The Farr assay probably has the highest specificity, and lowest sensitivity of the three assays for the diagnosis of SLE, while solid-phase assays have the highest sensitivity and lowest specificity; the crithidia assay falls in between. The Farr assay also has the disadvantage of using radioactive material, which most labs shun, and the crithidia test is very labor intensive. I prefer the ELISA assay for everyday use.
Anti-dsDNA are specific (95%) though not highly sensitive (70%) for SLE, making them very useful for diagnosis when positive.9 (See Table 2, p. 17.) They are occasionally found in other conditions, including rheumatoid arthritis, juvenile arthritis, drug-induced lupus, autoimmune hepatitis, and even in normal persons.
Titers of anti-dsDNA antibodies often fluctuate with disease activity, especially lupus nephritis, and are therefore useful in many patients for following the course of SLE. If a patient has a rising titer, or very high titer, but clinically is quiescent, I do not treat the serological abnormality, but consider it a warning sign that the patient needs to be followed more closely and treated when something happens clinically to warrant a change in therapy. However, some investigators have treated these serological abnormalities (especially when associated with low complement levels) and have reported improved clinical outcomes. Conversely, I use a falling titer in someone who is getting better after a flare on treatment as a guide in reducing treatment (e.g., corticosteroids and/or immunosuppressives).
The association between anti-dsDNA antibodies and other disease manifestations of SLE is far less clear. For example, there is no relationship between anti-dsDNA titer and disease activity of neuropsychiatric SLE.
Distinguishing active lupus manifestations from infectious complications or toxic effects of drugs—and from unrelated disease—is always a challenge. The presence of anti-dsDNA antibodies may be helpful in some patients in making this distinction.
Anti-Smith and Anti-Ribonucleoprotein Antibodies
Antibodies to Smith (Sm) and anti-ribonucleoprotein (anti-RNP) are most frequently detected by solid-phase immunoassays.24,27
Anti-Sm antibodies are found in only 10% to 40% of patients with SLE, but are very infrequent in patients with other conditions (i.e., they are not sensitive but are highly specific [see Table 2, p. 17]). Measurement of anti-Sm titers may be useful diagnostically, particularly at a time when anti-DNA antibodies are undetectable. Given the relatively low sensitivity of anti-Sm, however, a negative value in no way excludes the diagnosis of SLE.
Anti-RNP antibodies are found in about 40% to 60% of patients with SLE, but are not specific for SLE, being a defining feature of MCTD. These antibodies can also occur in low titers and low frequencies in other rheumatic diseases including RA and scleroderma. (See Table 2, p. 17.)
Neither the titer (levels) of anti-Sm nor anti-RNP antibodies correlates with any clinical activity.
Anti-Ro/SSA and Anti-La/SSB Antibodies
Antibodies to Ro/SSA and La/SSB are most frequently detected by solid-phase immunoassays.28,29 Anti-Ro/SSA and anti-La/SSB have been detected in high frequency in patients with Sjögren’s syndrome and in SLE, but also in patients with photosensitive dermatitis, and in 0.1% to 0.5% of healthy adults.
Anti-Ro/SSA antibodies are found in approximately 50% of patients with SLE. (See Table 2, p. 17.) They have been associated with photosensitivity, subacute cutaneous lupus, cutaneous vasculitis (palpable purpura), interstitial lung disease, neonatal lupus, and congenital heart block. Anti-Ro/SSA antibodies are found in approximately 75% of patients with primary Sjögren’s syndrome (see Table 2, p. 17), and high titers of these antibodies are associated with a greater incidence of extra glandular features, especially purpura and vasculitis. By contrast, Ro/SSA antibodies are present in only 10% to 15% of patients with secondary Sjögren’s syndrome associated with rheumatoid arthritis. Therefore, the presence of Ro/SSA or anti-La/SSB antibodies in patients with suspected primary Sjögren’s syndrome strongly supports the diagnosis.
Approximately 50% of patients with SLE who have anti-Ro antibody also have anti-La antibody, a closely related RNA-protein antigen. Similarly, most patients with Sjögren’s syndrome also have anti-La (SSB) antibodies. It is exceedingly rare to find patients with anti-La antibodies without anti-Ro antibodies.
In my opinion, the indications for ordering an anti-Ro/SSA and anti-La/SSB antibody tests are as follows:
- Women with SLE who are pregnant or may become pregnant in the future;
- Women who have a history of giving birth to a child with heart block or myocarditis;
- Patients with a history of unexplained photosensitive skin eruptions;
- Patients strongly suspected of having SLE but who have a negative ANA test; or
- Patients with symptoms of xerostomia, keratoconjunctivitis sicca, and/or salivary and lacrimal gland enlargement.
Antiribosomal P Protein Antibodies
Anti-ribosomal P protein antibodies have been detected in 10% to 20% of patients with SLE in the United States and 40% to 50% of Asian patients with SLE, but rarely in other rheumatic diseases.30 Anti-ribosomal P protein antibodies may be highly specific for lupus-associated psychosis, although not all studies have confirmed this. Depression and renal and liver involvement in patients with lupus have also been associated with anti-ribosomal P protein antibodies in some small studies.
Because of the low sensitivity and poor clinical correlation, many investigators—including myself—see little clinical usefulness in this test.
Anti-Centromere Antibodies
Anti-centromere antibodies (ACAs) are found almost exclusively in patients with limited cutaneous systemic sclerosis (lcSSc) especially in those with CREST.31 ACAs have been observed in 57% of patients with CREST but have also been seen in patients with other conditions, including in some patients with Raynaud’s phenomenon alone. ACAs are typically detected by the characteristic IF pattern on Hep-2 cells.
Anti–Scl-70 (topoisomerase-1) Antibodies
Approximately 15% to 20% of patients with scleroderma have antibodies to a 70kd protein (topoisomerase-1), subsequently named Scl-70.31 The usual method for detection is by ELISA. The presence of these antibodies appears to increase the risk for pulmonary fibrosis among patients with scleroderma and is quite specific for the disease.
Summary
If one suspects SLE based on the history, physical examination, and complete blood count (e.g., leucopenia, thrombocytopenia), request an ANA test. If it is negative, it is highly unlikely that the patient has SLE, and the patient can be reassured. If the titer is <1:160, SLE is unlikely, and the patient can be reassured that SLE is unlikely, but the patient should be followed periodically. A titer of ≥1:160 increases the likelihood of SLE and related disorders. However, it is important to remember that 85% of individuals with a positive ANA do not have SLE. If the ANA is positive, one should also test for antibodies to dsDNA, Sm, RNP, Ro/SSA, and La/SSB. The presence of these antibodies greatly increases the likelihood that the patient has SLE and related disorders (see Table 2, p. 17), and the patient needs to be followed more closely. The detection of anti-centromere and anti–Scl-70 antibodies is very useful for the diagnosis of scleroderma. The detection of antibodies to Ro/SSA and La/SSB are also useful for the diagnosis of Sjögren’s syndrome.
The only one of these tests that is useful to monitor activity is the anti-dsDNA.
Acknowledgement: I am indebted to the work of many authors of UpToDate in Medicine, whose work provided a useful framework for the development of this paper, as well as to Drs. Robert Shmerling, David Lee, and Donald Bloch, with whom I have written papers and chapters on this same subject.
Dr. Schur is professor of medicine at Harvard Medical School and the division of rheumatology, immunology, and allergy in the department of medicine at Brigham and Women’s Hospital in Boston.
References
- Reichlin M. Measurement and clinical significance of antinuclear antibodies. UpToDate. October 2008.
- American College of Rheumatology Ad Hoc Committee on Immunologic Testing Guidelines. Guidelines for immunologic laboratory testing in the rheumatic diseases: An introduction. Arthritis Rheum. 2002;47:429-433.
- Solomon DH, Kavanaugh AJ, Schur PH, et al. Evidence-based guidelines for the use of immunologic tests: Antinuclear Antibody testing. Arthritis Rheum. 2002;47:434-444.
- Sinclair D, Saas M, Williams D, Hart M, Goswami R. Can an ELISA replace immunofluorescence for the detection of anti-nuclear antibodies?—The routine use of anti-nuclear antibody screening ELISAs. Clin Lab. 2007;53:183-191.
- Avaniss-Aghajani E, Berzon S, Sarkissian A. Clinical value of multiplexed bead-based immunoassays for detection of autoantibodies to nuclear antigens. Clin Vaccine Immunol. 2007;14:505-509.
- Copple SS, Martins TB, Masterson C, Joly E, Hill HR. Comparison of three multiplex immunoassays for detection of antibodies to extractable nuclear antibodies using clinically defined sera. Ann N Y Acad Sci. 2007;1109:464-472.
- Caramaschi P, Ruzzenente O, Pieropan S, et al. Determination of ANA specificity using multiplexed fluorescent microsphere immunoassay in patients with ANA positivity at high titres after infliximab treatment: Preliminary results. Rheumatol Int. 2007;27:649-654.
- Biagini RE, Parks CG, Smith JP, Sammons DL, Robertson SA. Analytical performance of the AtheNA MultiLyte ANA II assay in sera from lupus patients with multiple positive ANAs. Anal Bioanal Chem. 2007;388:613-618.
- Gonzalez C, Garcia-Berrocal B, Perez M, et al. Laboratory screening of connective tissue diseases by a new automated ENA screening assay (EliA Symphony) in clinically defined patients. Clin Chim Acta. 2005;359:109-114.
- Eissfeller P, Sticherling M, Scholz D, et al. Comparison of different test systems for simultaneous autoantibody detection in connective tissue diseases. Ann N Y Acad Sci. 2005;1050:327-339.
- Smith J, Onley D, Garey C, et al. Determination of ANA specificity using the UltraPlex platform. Ann N Y Acad Sci. 2005;1050:286-294.
- Martins TB, Burlingame R, von Muhlen CA, et al. Evaluation of multiplexed fluorescent microsphere immunoassay for detection of autoantibodies to nuclear antigens. Clin Diagn Lab Immunol. 2004;11:1054-1059.
- Bernardini S, Infantino M, Bellincampi L, et al. Screening of antinuclear antibodies: Comparison between enzyme immunoassay based on nuclear homogenates, purified or recombinant antigens and immunofluorescence assay. Clin Chem Lab Med. 2004;42:1155-1160.
- Nossent H, Rekvig OP. Antinuclear antibody screening in this new millennium: Farewell to the microscope? Scand J Rheumatol. 2001;30:123-126; discussion 127-128.
- Lopez-Hoyos M, Rodriguez-Valverde V, Martinez-Taboada V. Performance of antinuclear antibody connective tissue disease screen. Ann N Y Acad Sci. 2007;1109:322-329.
- Ghillani P, Rouquette AM, Desgruelles C, et al. Evaluation of the LIAISON ANA screen assay for antinuclear antibody testing in autoimmune diseases. Ann N Y Acad Sci. 2007;1109:407-413.
- Bonilla E, Francis L, Allam F, et al. Immunofluorescence microscopy is superior to fluorescent beads for detection of antinuclear antibody reactivity in systemic lupus erythematosus patients. Clin Immunol. 2007;124:18-21.
- Nifli AP, Notas G, Mamoulaki M, et al. Comparison of a multiplex, bead-based fluorescent assay and immunofluorescence methods for the detection of ANA and ANCA autoantibodies in human serum. J Immunol Methods. 2006;311: 189-197.
- Shovman O, Gilburd B, Zandman-Goddard G, et al. Multiplexed AtheNA multi-lyte immunoassay for ANA screening in autoimmune diseases. Autoimmunity. 2005;38:105-109.
- Ulvestad E. Performance characteristics and clinical utility of a hybrid ELISA for detection of ANA. Apmis. 2001;109:217-222.
- Olaussen E, Rekvig OP. Screening tests for antinuclear antibodies (ANA): Selective use of central nuclear antigens as a rational basis for screening by ELISA. J Autoimmun. 1999; 13:95-102.
- Homburger HA, Cahen YD, Griffiths J, Jacob GL. Detection of antinuclear antibodies: Comparative evaluation of enzyme immunoassay and indirect immunofluorescence methods. Arch Pathol Lab Med. 1998;122:993-999.
- Gniewek RA, Stites DP, McHugh TM, Hilton JF, Nakagawa M. Comparison of antinuclear antibody testing methods: Immunofluorescence assay versus enzyme immunoassay. Clin Diagn Lab Immunol. 1997;4:185-188.
- Reichlin M. Antibodies to DNA, SM, and RNP. UpToDate. October 2008.
- Hahn BH. Antibodies to DNA. New Engl J Med. 1998;338:1359.
- Kavanaugh AF, Solomon DH, the American College of Rheumatology Ad Hoc Committee on Immunologic Testing Guidelines. Guidelines for immunologic laboratory testing in the rheumatic diseases: Anti-DNA antibody tests. Arthritis Rheum. 2002; 47:546-555.
- Benito-Garcia E, Schur PH, Lahita R. Guidelines for Immunologic laboratory testing in the rheumatic diseases: Anti-Sm and Anti-RNP antibody tests. Arthritis Rheum. 2004; 51:1030-1044.
- Reichlin M. Clinical significance of anti-Ro/SSA and anti-La/SSB antibodies. UpToDate. October 2008.
- Reveille JD, Sherrer YRS, Solomon DH, Schur P, Kavanaugh A. Evidenced-based guidelines for the use of immunological laboratory tests: Anti-Ro (SS-A) and La (SS-B): A report from the ACR Ad Hoc Committee on Immunological Testing Guidelines. Submitted for publication.
- Reichlin M. Antiribosomal P protein antibodies. UpToDate. October 2008.
- Denton CP. Classification of scleroderma. UpToDate. October 2008.