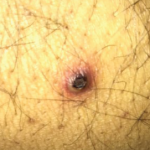
NEW YORK (Reuters Health)—Methotrexate appears to increase the risk of recurrent nonmelanoma skin cancer (NMSC) in patients with rheumatoid arthritis (RA), according to a retrospective study.
“We have known for some time that several of the immunosuppressive agents used to treat RA and inflammatory bowel disease (IBD) are also associated with an increased risk of de novo nonmelanoma skin cancer,” said Dr. Frank I. Scott from the University of Pennsylvania in Philadelphia.
“This is the first study to demonstrate that the risk of a second NMSC is also elevated when treating those that have already had an incident NMSC diagnosis, particularly with methotrexate, which is widely considered to be the backbone therapy in RA,” he told Reuters Health by email. “Further research is required examining the risks of anti-TNFs and thiopurines with a second NMSC.”
Methotrexate, thiopurines and anti-TNF drugs have been associated with hematologic and dermatologic malignancies, but there are limited data regarding the effect of these medications on the risk of cancer recurrence.
Dr. Scott and colleagues assessed the risk of a second NMSC in a retrospective study of Medicare beneficiaries with RA (n=6,672) or IBD (n=2,619) or both (n=169) exposed to methotrexate, thiopurines or biologic agents.
The incidence rate per 1,000 person-years of a second NMSC was 58.2 with RA and 58.9 with IBD, the team reports in JAMA Dermatology, online Oct. 28.
Among patients with RA, methotrexate use was associated with a 60% increased risk of a second NMSC. After adjusting for the use of other medications, the risk of a second NMSC rose by 10% with less than a year of methotrexate exposure, by 16% with one to two years of exposure, by 36% with two to three years of exposure, and by 59% with more than three years of exposure.
Short-term use of anti-TNF agents was associated with a 43% increase in risk of a second NMSC, but longer use didn’t appear to up the risk.
Among IBD patients, thiopurine and corticosteroid use was not associated with increased risk of a second NMSC.
“However, I would caution against assuming that anti-TNFs or thiopurines impart lesser risk, as it is quite possible that our non-significant results were driven by sample size in these smaller populations,” Dr. Scott said.
“The decision to use any medication with known associations with rare but serious adverse events should be made on a case by case basis, not only taking into account patients’ disease severity but also other comorbidities and individual preferences,” Dr. Scott concluded. “We are hopeful that these data will help guide clinicians in conversations about these risks with their patients.”