Results from a new study reinforce a longstanding concern that the immunosuppressant and biologic agents commonly used to treat patients with autoimmune disease may increase the risk of nonmelanoma skin cancer (NMSC).
Results from a new study reinforce a longstanding concern that the immunosuppressant and biologic agents commonly used to treat patients with autoimmune disease may increase the risk of nonmelanoma skin cancer (NMSC).
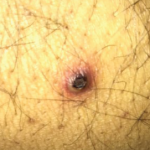
NMSC, also known as squamous cell carcinoma, is the most frequently diagnosed malignant neoplasm in the U.S. Unfortunately, immunosuppressive therapy is a known risk factor for NMSC. In particular, methotrexate is associated with an increased risk of NMSC in patients with a history of NMSC. Patients with rheumatoid arthritis (RA) and NMSC also have an increased risk for NMSC recurrence when treated with anti-tumor necrosis factor (anti-TNF) and methotrexate.
Frank I. Scott, MD, MSCE, assistant professor of medicine at the University of Pennsylvania in Philadelphia, and colleagues questioned whether immunosuppressive treatment regimens were associated with an increased risk for NMSC. The results of their retrospective cohort study were published online on Oct. 28 in JAMA Dermatology.1 The team studied claim-based data from patients with RA and inflammatory bowel disease (IBD) who experienced NMSC, as identified by a combination of diagnostic and procedural codes. They then evaluated the effect of immunosuppressive therapies on the risk of NMSC recurrence.
Methotrexate Increases Risk for NMSC
The investigators identified 6,841 patients with RA, 2,788 patients with IBD and 169 patients with both RA and IBD. A total of 1,291 individuals had NMSC recurrence, yielding an incidence rate of NMSC recurrence of 58.2 per 1,000 person-years in patients with RA and 58.9 per 1,000 person-years in patients with IBD. They calculated that one year or more of methotrexate use (once adjusted for other medications) was associated with an increased risk of NMSC (hazard ratio: 1.24; 95% confidence interval, 1.04–1.48). The median time of methotrexate exposure between the first NMSC and recurrence was 1.64 years. The researchers also observed an increased risk of NMSC with increased duration of methotrexate exposure. The increase was even greater (34 per 1,000 person-years) when methotrexate was used with non-immunosuppressive therapies.
In contrast, patients who were treated with methotrexate for RA did not experience a further increase in risk for NMSC recurrence when treated with abatacept or rituximab. However, in the case of patients with RA who were treated with methotrexate and anti-TNFs, the incidence of NMSC recurrence increased significantly, by 19 per 1,000 person-years. Anti-TNF therapy alone was also significantly associated with increased risk for NMSC recurrence in patients with RA. Unfortunately, although the investigators were interested in exploring the effects of such drugs as leflunomide, which were prescribed to these patient populations, the sample size was too small to perform an analysis.