Tefi / shutterstock.com
Genes may predispose people to have certain microbial signatures in their gut that, in turn, make them susceptible to developing reactive arthritis. This is the main finding of a recent study in which researchers investigated whether perturbations in the intestinal microbiome play a role in susceptibility to reactive arthritis in the face of triggers, such as gastrointestinal (GI) or genitourinary (GU) infection.1
The study, “Gut Microbiota Perturbations in Reactive Arthritis and Postinfectious Spondyloarthritis” by Julia Manasson, MD, rheumatology post-doctoral fellow, Division of Rheumatology, Department of Medicine, NYU Langone Health, New York, and colleagues, is the first to use culture-independent, high-throughput DNA sequencing to examine the gut microbiome in patients with reactive arthritis and postinfectious spondyloarthritis.
“Although reactive arthritis is thought to be triggered by certain GI or GU infections, we often do not know in an individual patient what organisms may be responsible,” says Allen Steere, MD, professor of medicine, Harvard Medical School, director of clinical research, Rheumatology Unit, Massachusetts General Hospital, Boston, who commented on the study for The Rheumatologist.
By using a new, DNA sequencing, discovery-based method to determine the range of organisms present in the patients’ stool samples, the investigators found “a broader range of organisms than was previously known in reactive arthritis,” he says, adding that the results “support the hypothesis that certain organisms in the gut may shape immune responses that are important to reactive arthritis.”
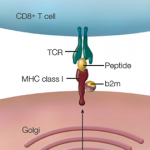
The study also looked at the relationship between host genes and microbiota, and particularly focused on the human leukocyte antigen (HLA) genes. Previous data have shown an association between HLA-B27 and reactive arthritis, although the presence of HLA-B27 does not guarantee the development of disease. Further, HLA-B27 is highly prevalent in Caucasian Northern Europeans, but absent in many other populations in whom reactive arthritis develops.
The current study included patients from Guatemala, an area where there is a low prevalence of the HLA-B27 allele. Only four patients in the study were HLA-B27 positive. The study found that patients who were HLA-A24 positive showed distinct gut microbiota profiles compared with those who were HLA-A24 negative, irrespective of disease status. This was also true in the case of HLA-B35. This finding suggests host genetics contributes to the richness and diversity of gut microbiota.
“Genetics seems to play a role in determining gut microbiota composition,” says lead author Dr. Manasson. “This, in turn, may confer susceptibility to developing disease, [such as] reactive arthritis, when in the presence of other triggers.”
Study Highlights
As mentioned above, the study looked at two main issues to understand the role of gut microbiota in the pathogenesis of reactive arthritis. The first issue was to characterize the types of bacteria present in the gut of patients who develop reactive arthritis/postinfectious spondyloarthritis compared with controls who were exposed to the same types of infections but did not develop disease. The second issue was to characterize the relationship of host genes to microbiota.

Dr. Manasson
To do this, Dr. Manasson and colleagues focused on 64 adult subjects who all had a previous GI or GU infection. Of these, 32 subjects subsequently developed reactive arthritis/postinfectious spondyloarthritis and 32 did not. As mentioned previously, all participants were from Guatemala, a geographical region where HLA-B27 prevalence is low.
All participants were between the ages of 18 and 55 years old and had experienced a GI, GU or sexually transmitted infection three to six months prior to study enrollment. Patients were excluded from the study if they were diagnosed with inflammatory bowel disease, psoriasis, other inflammatory arthritis or autoimmune disease, or active cancer.
Each participant underwent a clinical examination that included a history and physical examination, as well as ultrasound. Data collected included HLA typing and stool for microbiota analysis.
The study found that gut microbial richness and diversity were similar between both groups of patients, but that participants with reactive arthritis had significantly greater abundance of enteropathogens than controls. Specifically, the study found a significantly higher abundance of bacteria from the genus Erwinia, which is commonly linked to more than 97% of the gut bacteria, associated with reactive arthritis. These bacteria include Salmonella, Shigella and Yersinia. The study also found a higher abundance of the pathogen Pseudomonas in the participants who developed reactive arthritis than in controls.

Dr. Steere
In addition, the study found several gut commensals were decreased in the patients with reactive arthritis compared with controls. Specifically, control subjects were enriched in the Lachnospiraceae taxa, including Blautia, Coprococcus, Roseburia and Collinsella.
“Our study showed that the abundance of enteropathogens was increased and the abundance of commensal organisms was decreased in subjects who developed reactive arthritis/postinfectious spondyloarthritis compared to those who did not,” says Dr. Manasson, “which may suggest a susceptibility to disease development.”
Overall, Dr. Manasson says the study showed host genes were associated with particular microbial signatures and that differences in microbiota composition were found between patients who developed reactive arthritis and controls. “We believe that genes may predispose individuals to certain gut microbial signatures, which, in the right setting, may make individuals more or less susceptible to disease after GU or GI infections,” she says.
Future Research
Going forward, Dr. Manasson and colleagues emphasize the need for further research to close the gaps that persist in better understanding the interactions between gut microbiota, genetics and susceptibility of postinfectious chronic inflammatory conditions.
Saying these findings contribute to a better understanding of the environmental factors important to the development of various types of chronic arthritis, Dr. Steere emphasizes they do not yet affect how these conditions are diagnosed or treated.
“The long-range hope is that tests can be developed to help us identify a patient with reactive arthritis and that treatment directed against causative organisms, as well as suppression of immune responses, may be more effective therapy,” he says.
Mary Beth Nierengarten is a freelance medical journalist based in Minneapolis.
Resources
- Manasson J, Shen N, Garcia Ferrer HR, et al. Gut microbiota perturbations in reactive arthritis and postinfectious spondyloarthritis. Arthritis Rheumatol. 2018 Feb;70(2):242–254.