Dr. Esdaile
Dr. Esdaile, who trained nearby at Yale University, spoke to Dr. Rothfield in detail about the work. He coordinated the study out of Montreal General Hospital, Quebec, Canada, where he was working at the time. He recruited doctors from two other hospitals in Montreal, from a hospital in Ottawa and from another in Hamilton, Ontario. Those physicians then asked their colleagues to send them any lupus patients who were doing well and currently taking hydroxychloroquine.
“The fact that the Canadians banded together to do this is a remarkable substory of this paper,” says Dr. Petri. “They are lumped together as the Canadian Hydroxychloroquine Study Group. This is a great example of clinicians caring about lupus and not caring about their own names or their own egos.”
Study Design & Outcomes
Dr. Esdaile and colleagues opted to perform a prospective, double-blind, randomized withdrawal study to examine the potential protective role of hydroxychloroquine in preventing disease flares.
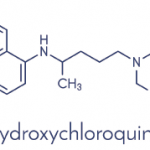
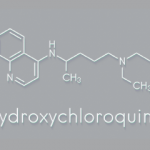
The group studied lupus patients who had been taking hydroxychloroquine in doses ranging from 100 mg to 400 mg per day. The average length of time the lupus patients had been taking hydroxychloroquine was three years, and all patients had taken it for at least six months. All had experienced clinical remission or only minimal disease activity for the three months prior to study initiation. The patients were randomized to either continue taking hydroxychloroquine at their current dose or to take a placebo.1
Dr. Esdaile explains the characteristics of lupus make it challenging to study. He partly chose the withdrawal design because lupus often requires intensive intervention with stronger drugs, such as prednisone for flares of disease activity. He believed the effect of that would obscure the impact of hydroxychloroquine, making benefits harder to see.
In 2016, the American Academy of Ophthalmology published guidelines for hydroxychloroquine patient monitoring, recommending optical coherence tomogram at baseline, five years thereafter & then every year following for patients without major risk factors for retinopathy. … In terms of toxicity, recent guidelines … recommend lowering the dosage from 6.5 mg/kg patient weight to less than 5.0 mg/kg.
“If you used people who had never seen hydroxychloroquine, the flare would create a nightmare for analysis,” he says. “Whereas the way we designed it, the flareup was the outcome.” Additionally, because many doctors and patients had concerns about taking the drug continuously, Dr. Esdaile says it would have been more difficult to find patients to enroll in a trial of patients naive to hydroxychloroquine.
Blinding the study properly was a challenge, because hydroxychloroquine formulations at the time had a strong, unpleasant taste. Dr. Esdaile worked with the drug manufacturer to obtain a drug and a placebo in identical shapes. To help with the disguise, the placebos were coated with a trace amount of hydroxychloroquine (<1 mg), so they would also have an unpleasant taste.
The group assessed patients every four weeks over a 24-week period using standard clinical assessments and laboratory tests. Final analyses included 47 patients with lupus.1 Using data from Dr. Rothfield’s work, Dr. Esdaile and colleagues estimated that staying on hydroxychloroquine would reduce flares by about 50% with their study design.
Dr. Esdaile and colleagues found the relative risk of a clinical flare-up (the primary outcome) was 2.5 times higher in patients taking placebo than in those who had continued to take hydroxychloroquine. Moreover, the relative risk of the secondary outcome, severe disease exacerbation, was six times higher in patients taking placebo than in those who had continued taking hydroxychloroquine. (This, however, did not reach statistical significance, which was unsurprising given study size and length.)
The findings also provided early evidence that hydroxychloroquine treatment might reduce nephritic flares successfully. Efforts to properly blind the study were successful, with only 53% of participants correctly guessing whether they were taking hydroxychloroquine or placebo.1
“It was a remarkably simple study, and the preventive role of hydroxychloroquine was so strong,” Dr Petri remarks. She contrasts the sample size with those found in modern studies using much higher numbers of patients. She adds, “I often say if something really works, you don’t need a lot of patients to show it. And that’s one reason why I think this is such a wonderful study.”
The randomized withdrawal trial design is now only rarely used in lupus, although it is still sometimes employed in juvenile chronic arthritis studies. Dr. Petri says, “I think this shows that maybe we’ve forgotten it’s a good study design.”
Randomized withdrawal trials do present problems of generalizability. For example, such a trial design excludes patients who had previously discontinued hydroxychloroquine due to lack of response, toxicity or other factors, and thus may tend to overestimate efficacy.
But Dr. Esdaile emphasizes that one of the design’s strengths is the ability to show an effect with a smaller sample size.