In the group receiving 300 mg mepolizumab monthly, 28% were in remission for at least 24 weeks in the 48-week study, compared with only 3% in the placebo group; similarly, 32% of patients in the mepolizumab group were in remission at both Weeks 36 and 48 (prespecified), compared with 3% in the placebo group. However, to dampen enthusiasm somewhat, 47% of patients receiving mepolizumab never met criteria for remission (compared with 81% in the placebo group), and the average prednisone dose dropped only from 12 mg to 5 mg (compared with being unchanged at 10–12 mg in the placebo group throughout the trial). It is also impossible to determine how many patients had flares of vasculitis (i.e., polyangiitis) or severe parenchymal eosinophilic disease above and beyond asthma or sinonasal disease, because vasculitis was defined as any items on the Birmingham Vasculitis Activity Score (BVAS), which includes such factors as fatigue and arthralgia.
Because rituximab is as effective as cyclophosphamide for treating ANCA-positive patients with GPA and MPA, the question arises whether rituximab is also effective in treating EGPA, which also features small-vessel vasculitis and in which 30–40% of patients have ANCA, usually anti-myeloperoxidase. This question has not yet been addressed in a trial, but experience suggesting benefit has been described in a retrospective study of 41 patients assembled from several referral centers.12 Historical manifestations, prior disease courses and prior treatments were diverse, similar to large case series, and 44% of patients were ANCA positive. Whether glucocorticoids were increased in some patients around the time rituximab was initiated was not reported, but the median dose of prednisone/prednisolone dropped from 15 mg at baseline to 8 mg at six and 12 months after starting rituximab. Remission, defined as BVAS=0, was achieved in 12 of 15 (80%) ANCA-positive patients and eight of 21 (38%) ANCA-negative patients.
Although [Puechal] does not really show that azathioprine doesn’t work in EGPA, it strongly suggests that it doesn’t work very well.
What I Do … (Is 30 Ever a Lot?)
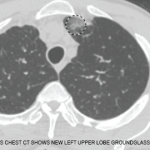
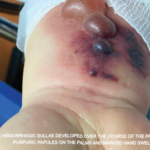
For assessing severity, I find the original FFS (including CNS, cardiac, significant renal or severe GI involvement) to be useful in defining severe disease, but I would add pulmonary hemorrhage (rare in EGPA) and have a low threshold for adding peripheral neuropathy in light of its likelihood of leading to chronic pain and disability.
For patients with severe disease, I use a regimen similar to GPA or MPA, except that for EGPA I would almost always use cyclophosphamide (oral or IV, for three to six months) rather than rituximab. In contrast to GPA and MPA, for ANCA-positive patients with EGPA I would count on rituximab only to control GN or pulmonary hemorrhage. The glucocorticoid regimen I typically use in patients with severe EGPA, as with GPA or MPA, is an IV pulse of methylprednisolone, followed by 60, 50, 40, 30, 20, 15, 10, 7.5, 5 and 2.5 mg for two weeks each. In practice, the prednisone tapering usually needs to be slowed and eventually halted based on recurrent inflammatory disease in the upper or lower airway. Then, the task is steroid sparing, just as in a patient whose initial presentation was nonsevere.