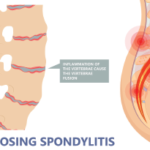
NSAIDs are clinically effective and considered a first-line therapy in patients with AS, Dr. Ritchlin said. “NSAIDs also may reduce radiographic progression in at-risk patients,” he added. “Studies have not been done to demonstrate if specific NSAIDs are more favorable than others, but continuous use of these medications at full dose is recommended based on data from recent clinical studies.”
A drug that Dr. Ritchlin said shows promise in PsA for inhibition of radiographic damage, aside from TNFi, is ustekinumab, an IL-12/23 p40 inhibitor. Results from the combined radiographic data from the PSUMMIT1 and two studies indicated that ustekinumab reduced X-ray progression of structural damage in PsA at Week 24 compared with placebo.
In patients with AS who had active disease despite NSAID therapy, ustekinumab treatment was effective in significantly reducing signs and symptoms of disease. Patients in this small, open-label study received 90 mg ustekinumab subcutaneously at baseline, Week 4 and Week 16.
“The bony changes in the spine and peripheral joints have been a major burden for SpA patients,” Dr. Ritchlin said. “New data have unveiled potential novel targets, and older drugs, such as NSAIDs, can decrease pathologic new bone formation. Animal models suggest a central role of IL-23 and IL-22 in osteoproliferation, but confirmatory studies in humans are required to support this paradigm. Early diagnosis and treatment of patients with SpA will likely limit new bone formation and result in improved outcomes.”
Kimberly J. Retzlaff is a medical journalist based in Denver.
References
- Hreggvidsdottir HS, Noordenbos T, Baeten DL. Inflammatory pathways in spondyloarthritis. Mol Immunol. 2014;57(1):28–37.
- Lories RJ, McInnes IB. Primed for inflammation: Enthesis-resident T cells. Nat Med. 2012;18(7):1069–1076.
- Jacques P, Lambrecht S, Verheugen E, et al. Extended report: Proof of concept: Enthesitis and new bone formation in spondyloarthritis are driven by mechanical strain and stromal cells. Ann Rheum Dis. 2014;73:437–445.