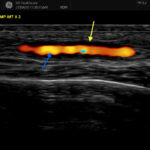
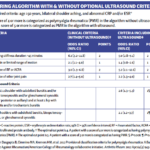
Background/Purpose It has been reported that 20–50% of patients with polymyalgia rheumatica (PMR) have subclinical giant cell arteritis (GCA). The natural history of ultrasound-defined subclinical GCA in PMR is not known. Methods Twenty-five newly diagnosed PMR patients who met a clinical diagnosis for PMR, verified by two rheumatologists, were examined by ultrasound. All six branches…